This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Tuberculosis vaccine does not protect elderly against COVID-19, finds large Dutch study
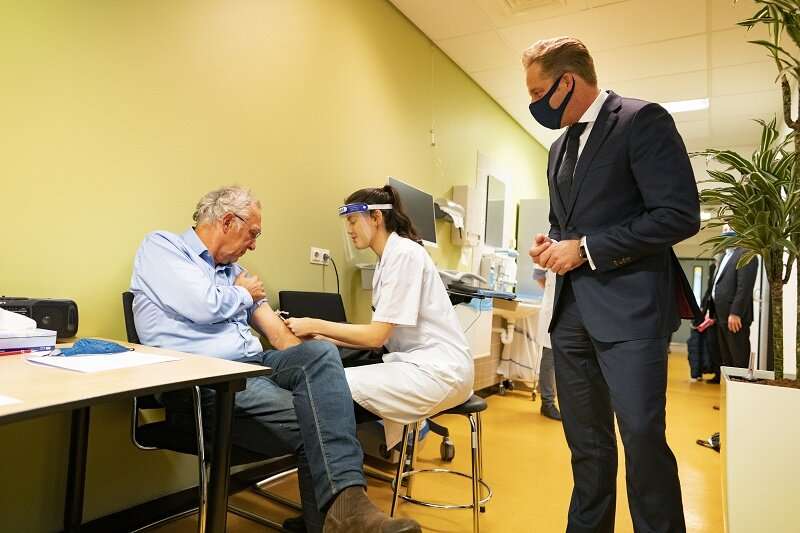
The tuberculosis vaccine (or BCG vaccine) does not protect the elderly with co-morbidities against disease symptoms caused by a coronavirus infection. This was reported in the BCG-PRIME study which was initiated during the early days of the coronavirus pandemic when no vaccines against the virus were available. As a collaborative effort, the study was designed and conducted in a very short time in 20 Dutch hospitals led by UMC Utrecht. After the main findings were released in 2021, the full results of the study were published this week in Clinical Microbiology and Infection.
In the early phase of the coronavirus pandemic, there was a major medical and societal need for a vaccine that could protect against COVID-19. This applied in particular to elderly people with comorbidities because it had quickly become clear that they were at increased risk of a severe course of the infection.
One candidate drug to bridge the period to effective COVID-19 vaccines and antivirals was the Bacillus Calmette-Guérin (BCG) vaccine. This vaccine—the most widely administered vaccine in the world—has been used in many countries for decades to prevent tuberculosis (TB). The BCG vaccine has the additional advantage of inducing a defense through the innate immune system that is broader than just preventing TB. Moreover, a few small studies had indicated that this vaccine could potentially protect elderly people from respiratory infections—particularly infections from a viral origin.
The hypothesis that the BCG vaccine might protect against severe COVID-19 led Dutch researchers guided by UMC Utrecht to urgently set up a large, well-controlled study that could provide a definitive answer to the question of whether the BCG vaccine protects elderly people with co-morbidities against COVID-19. The study was conducted from September 2020 onwards at 20 Dutch hospitals, including all UMCs and the Santeon top clinical hospitals.
BCG vaccine offers no protection
Investigator and first author of the article Eva Koekenbier MD (Program Epidemiology of Infectious Diseases, UMC Utrecht) summarizes the results of the study: "The main outcome of the BCG-PRIME study was the occurrence of COVID-19. The analysis showed that disease manifestations of COVID-19 occurred as frequently (hazard ratio 1.12; 95% confidence interval 0.87–1.44) in elderly people with co-morbidities who had received the BCG vaccine (129 out of 3,058 participants) as in those who had received placebo (115 out of 3,054 participants). We also found no difference between the two groups in terms of the number of hospitalizations due to COVID-19, the number of respiratory infections requiring medical treatment, the number of serious adverse events and mortality."
Study lead Prof. Marc Bonten MD Ph.D. (Program on Epidemiology of Infectious Diseases, UMC Utrecht) added, "Despite the fact that the outcome of our study was disappointing, we can be proud of the fact that we were able to successfully design and conduct the study—the largest to date on this topic—in a very short time and under great pressure. After only 4 months, we were able to conclude from a preliminary analysis that there was no major protective effect, which is certainly of interest for future studies.
"Furthermore, this study is an excellent example of a special collaboration in which the clinical trial infrastructure at the UMCs, the SANTEON hospitals and at a number of other hospitals was efficiently used in order to scale up quickly, and allowing us to vaccinate the required number of participants in the shortest possible time."
The study
A total of 6,112 elderly with co-morbidities aged 60 years and older participated in the placebo-controlled BCG-PRIME study between September 2020 and December 2021. Participants were recruited through hospital wards, outpatient clinics and anticoagulation services. Patients were monitored for six months after vaccination via their own hospital.
The study had two primary endpoints: (a) the number of cases of proven COVID-19, or (b) the number of cases of respiratory tract infections for which medical treatment took place. Interim results were released through a press release in January 2021.
More information: Bacillus Calmette-Guérin vaccine for prevention of COVID-19 and other respiratory tract infections in older adults with comorbidities: a randomized controlled trial, Clinical Microbiology and Infection (2023). DOI: 10.1016/j.cmi.2023.01.019 , www.clinicalmicrobiologyandinf … (23)00044-7/fulltext