This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
What happens if we run out of antibiotics?
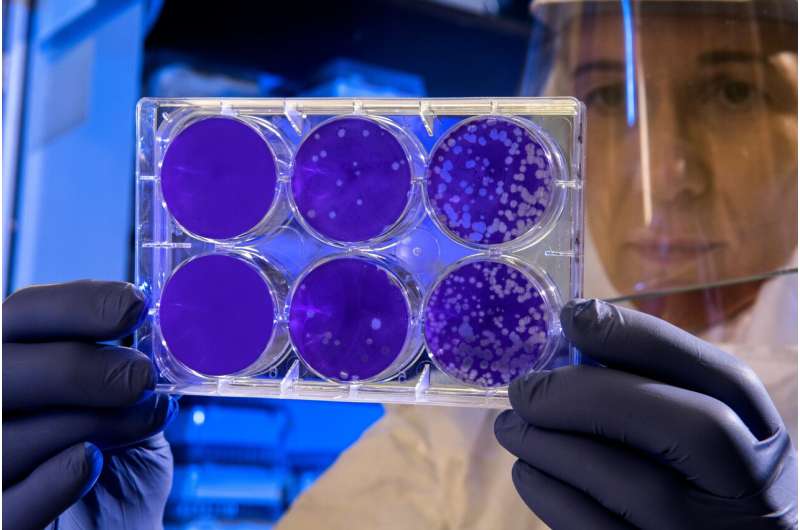
Widespread use of antibiotics in humans and animals has created growing resistance to treatments. What will happen if nothing is done? We asked expert Piotr Garstecki.
"With too few antibiotics under development to keep up with the rise of antibiotic-resistant infections, the world is starting to run out," says Garstecki, CEO at BacterOMIC.
This increasing resistance of microorganisms to antibiotics will result in the depletion of therapeutic options, a greater risk of bacterial spread, and prolonged infections, says Garstecki. This in turn will bring increased treatment costs, as well as higher mortality.
In fact, the World Health Organization predicts that worldwide death rates from drug-resistant infections will rise from 700,000 per year to 10 million by 2050, which would make them the main cause of death in humans.
The off-label use of antibiotics mainly concerns viral diseases, which—as an example—are the cause of about 80% of acute respiratory tract infections.
Overuse is one problem, but so is a downplaying of the problem due to a lack of awareness from both doctors and patients that each antibiotic may one day stop working, says Garstecki.
One potential unforeseen consequence if we do not act is a potential pandemic from a pathogen currently being kept in check by antibiotics, he explains. These include bacteria strains such as Staphylococcus aureus, which causes skin infections, Clostridium difficile, which can cause diarrhea and lead to sepsis—a life-threatening reaction to an infection, and Streptococcus pneumoniae, a bacterium that causes pneumonia, which can lead to hearing loss, brain damage and death.
"We definitely need targeted or—precision—antibiotic therapies, and for this we need precise and complete information about the susceptibility to antibiotics of the particular bacterial pathogen that causes infection," adds Garstecki.
Precision testing
In the BacterOMIC project, Garstecki's team developed an automated precision testing machine to perform mass screening for bacterial resistance. This tool can improve precision targeted medicine, in which treatments are allocated based on the susceptibility of a particular pathogen.
There are currently manual methods to screen for pathogen resistance, but BacterOMIC's innovative testing device delivers comprehensive antimicrobial susceptibility information in a single test—bringing savings in both cost and time.
A challenge requiring global effort
Another potential threat from an inadequate response might be a rising spread of viruses or zoonotic bacteria—the SARS-CoV-2 pandemic being one example.
"People are increasingly interfering with the natural environment and creating conditions for the spread of diseases, for example through migration and travel," explains Garstecki. Climate change, urbanization and dense population are also great breeding grounds for new pathogens, he adds.
"We have to tackle antimicrobial resistance now, or in the coming years we could find ourselves facing a new pandemic of sorts with serious drug-resistant infections impacting people on multiple continents," he says. "There is a clear and urgent need for new antibiotics and for better informed antibiotic stewardship that can only be guided by comprehensive diagnostics of antibiotic susceptibility of bacterial pathogens."