This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Breaking barriers in hepatitis C diagnosis and treatment for populations at risk
A study with people who inject drugs evaluated a minimally invasive test based on dried blood spots (DBS) for the monitoring of hepatitis C virus (HCV) infection. The use of DBS samples for HCV RNA detection and genotyping was shown to effectively assess cure after treatment and to differentiate between reinfection and treatment failure.
The results support the viability of decentralizing treatment and post-treatment monitoring for people who inject drugs, who frequently face challenges accessing the healthcare system. The study, which has been published in the Journal of Medical Virology, was carried out as part of a project with support from the "Conquering Hepatitis Via Microelimination" (CHIME) program.
Investigators from various research institutions collaborated in the project, including the Clinical Virology and New Diagnostic Tools research group, led by Dr. Elisa Martró, at Germans Trias i Pujol Research Institute (IGTP) and Dr. Sabela Lens from Hospital Clínic's Viral Hepatitis Group.
Towards elimination of hepatitis
In line with the strategy proposed by the World Health Organization for the elimination of viral hepatitis as a public health threat by 2030, and the Plan for Prevention and Control of Hepatitis in Catalonia, which Dr. Martró actively participates in, her group has been focused for years on simplifying the diagnosis of hepatitis C by developing and validating an assay which can detect the virus RNA using DBS samples.
These minimally invasive samples can be collected at harm reduction centers or drug dependence care and follow-up centers (known as CAS in Catalan), improving access to hepatitis C diagnosis for vulnerable populations, such as people who inject drugs. While this new test has demonstrated good clinical performance as a diagnostic tool for detecting HCV RNA before treatment in previous studies by the Clinical Virology and New Diagnostic Tools research group, the use of DBS samples had not been evaluated as a test for cure or for detecting reinfection after treatment.
A multidisciplinary research group has been able to pursue a project with a new model of care for hepatitis C, based on point-of-care diagnosis, treatment, and reinfection follow-up at the REDAN La Mina harm reduction center.
Since 2019, approximately 750 individuals who inject drugs have been tested though this initiative, which was designed by Dr. Sabela Lens from Hospital Clínic's Viral Hepatitis Unit, in collaboration with the Clinical Virology and New Diagnostic Tools Research Group at Germans Trias i Pujol Research Institute (IGTP), led by Dr. Martró from the Microbiology Service (LCMN) of the Germans Trias i Pujol Hospital (HUGTiP), as well as CEEISCAT and the Public Health Agency of Catalonia.
A model of decentralized care
In this project, Dr. Martró's group aimed to evaluate the clinical performance of a previously developed HCV-RNA assay based on DBS, for the assessment of cure and the detection of recurrent viremia after on-site treatment at the harm reduction center, compared to the commercially available HCV-RNA point-of-care test. Furthermore, they sought to assess the possibility of distinguishing between reinfection and treatment failure through HCV genotyping from baseline and follow-up DBS samples.
Typically, these assessments (cure and reinfection) are performed using venipuncture blood samples collected at healthcare centers, which can be difficult for people who inject drugs and have often limited access to the healthcare system. The recently published results demonstrate how the collection of DBS samples before and after treatment can simplify these assessments in decentralized test-and-treat programs.
"The success of the CHIME project lies in the decentralized diagnosis and treatment provided at REDAN La Mina. A nurse trained in hepatology assessments was included in the study to enroll and visit participants. The hepatologists at Hospital Clínic also reviewed each case and prescribed decentralized treatment. Additionally, Dr. Martró's group carried out HCV detection and sequencing from DBS samples collected before and after treatment. This pilot program involves HCV diagnosis on-site in less than an hour, treatment at the same center, and follow-up to assess reinfection," states Dr. Lens.
Detection made easier
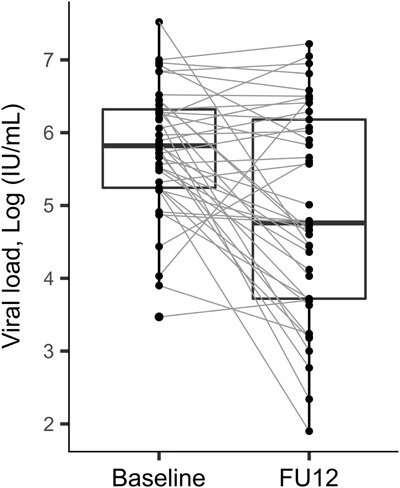
Reinfection is common in people who inject drugs and must be treated to prevent further transmission of the virus. During early reinfection, low levels of the virus may be present, making its detection in DBS samples challenging, as they only contain a small amount of blood. Of the 193 DBS samples tested after treatment, the DBS-based assay showed 100% specificity and sensitivity ranging from 84% to 96% based on different relevant viral load cut-offs, and similar rates as a test of cure (three months after treatment).
It must be born in mind that among the patients with recurrent viremia after treatment, one tenth had low viral loads. Moreover, HCV genotyping allowed researchers to classify 73% of viremic cases as either reinfection or treatment failure.
Collection of DBS samples was done before antiviral treatment and after treatment if recurrent viremia was detected by the commercially available point-of-care assay. Anna Not, the first author of the article (which will be part of her Ph.D.), explains that it "the use of DBS allowed us to sequence the virus before and after treatment and compare the sequences to determine if the virus was the same (indicating a treatment failure) or if it was different (indicating reinfection). This information enabled the hepatologist to decide on the most appropriate antiviral combination for the second treatment."
The research shows the potential of using DBS samples for determining cure and differentiating between reinfection and relapse after antiviral treatment for hepatitis C in people who inject drugs. The use of DBS samples makes it possible to decentralize treatment and follow-up, improving access to care for these people. Even so, Dr. Martró points out that "a small number of patients had low viral loads, which can hinder the detection of viremia and genotyping in DBS. As a result, repeat testing (e.g. every six months) is advised for individuals who are at risk of HCV reinfection."
More information: Anna Not et al, Usefulness of dried blood spot samples for monitoring hepatitis C treatment outcome and reinfection among people who inject drugs in a test‐and‐treat program, Journal of Medical Virology (2023). DOI: 10.1002/jmv.28544