This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
A novel blood serum assay to diagnose neurodegenerative diseases
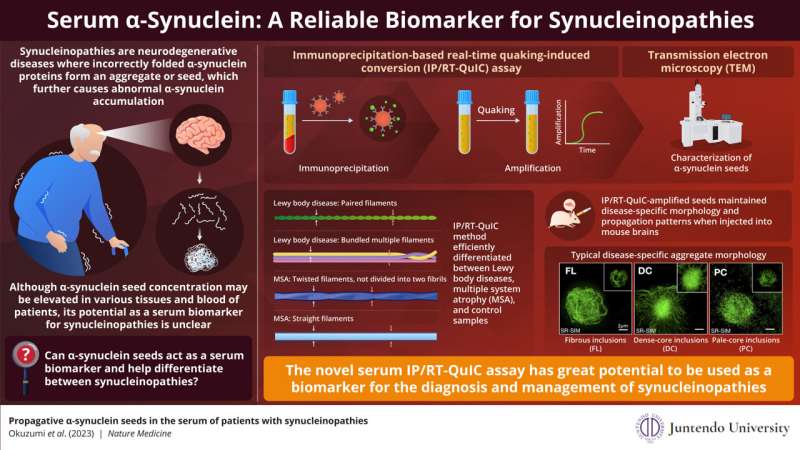
Synucleinopathies are a group of neurodegenerative diseases caused by the abnormal accumulation α-synuclein, a protein normally found in the brain and neurons. Incorrect folding of α-synuclein leads to formation of "seeds," which attract more α-synuclein proteins to form larger clumps. Although, α-synuclein seeds have been found in various tissues and blood of patients with synucleinopathies, its potential as a biomarker is ambiguous.
Recently, in a study published in Nature Medicine, Associate Professor Ayami Okuzumi along with Senior Associate Professor Taku Hatano, both from the Juntendo University School of Medicine, Senior Assistant Professor Gen Matsumoto at the Nagasaki University School of Medicine, and Professor Nobutaka Hattori from Juntendo University Faculty of Medicine /RIKEN Center for Brain Science, present a novel assay that can efficiently detect α-synuclein seeds from a patient's serum.
In this assay, named immunoprecipitation-based real-time quaking-induced conversion (IP/RT-QuIC), the α-synuclein seeds are isolated from the patient's serum through immunoprecipitation (protein separation using an antibody binding only to the target protein) followed by rapid amplification by real-time quaking-induced conversion (amplification induced by vigorous shaking). This method is highly sensitive, as it can detect serum α-synuclein seed concentrations as small as 1000pg/ml. This comes as great news since most existing diagnostic methods require cerebrospinal fluid for synuclein detection.
Sharing the objective of their study, Professor Hattori and his team explains, "In this study, we validated the usefulness of our novel assay system, IP/RT-QuIC, as a diagnostic marker of synucleinopathies. We propose that the fibril morphology of serum α-synuclein seeds and aggregates derived by IP/RT-QuIC can discriminate between Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA)."
The research team demonstrated that IP/RT-QuIC detected α-synuclein seeds efficiently in patients with neurodegenerative diseases and could distinguish them from people without degenerative diseases (controls). Next, they studied the structural properties of the amplified seeds using transmission electron microscopy (TEM).
They observed that the synuclein seed structure varied with the type of synucleinopathy. PD and DLB seeds showed paired filaments whereas MSA seeds had two distinct structures—twisted and straight filaments. This finding was confirmed that IP/RT-QuIC coupled with TEM can differentiate between synucleinopathies based on disease specific seed structure.
Further, when the researchers transduced amplified seeds into the HEK293T cell line stably expressing GFP-fused human α-synuclein with p.A53T mutation (in vitro) and injected seeds into mouse brains (in vivo), the seeds retained their aggregate forming capacity and diseases-specific seed structure. These aggregates displayed different morphologies depending on the disease type. Thus, specific synucleinopathies can be diagnosed by IP/RT-QuIC from the structural differences of the α-synuclein seeds and their aggregates.
This technique could help provide a quick and efficient diagnosis to patients. Professor Hattori and his team explains, "At present, a neurologist's consultation is necessary to diagnose synucleinopathies. However, using IP/RTQuIC, a general internist can make the diagnosis. Therefore, more patients with synucleinopathies may be diagnosed with precision and could receive appropriate treatment at an earlier stage."
The authors conclude with their future vision, "Our new IP/RT-QuIC assay may have many future applications as a biomarker for precise diagnosis and monitoring of treatment of neurodegenerative diseases in clinical trials. This simple diagnostic method will enable establishment of personalized therapy options for synucleinopathies."
More information: Nobutaka Hattori, Propagative α-synuclein seeds as serum biomarkers for synucleinopathies, Nature Medicine (2023). DOI: 10.1038/s41591-023-02358-9. www.nature.com/articles/s41591-023-02358-9