This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Double testing better at identifying bowel cancer
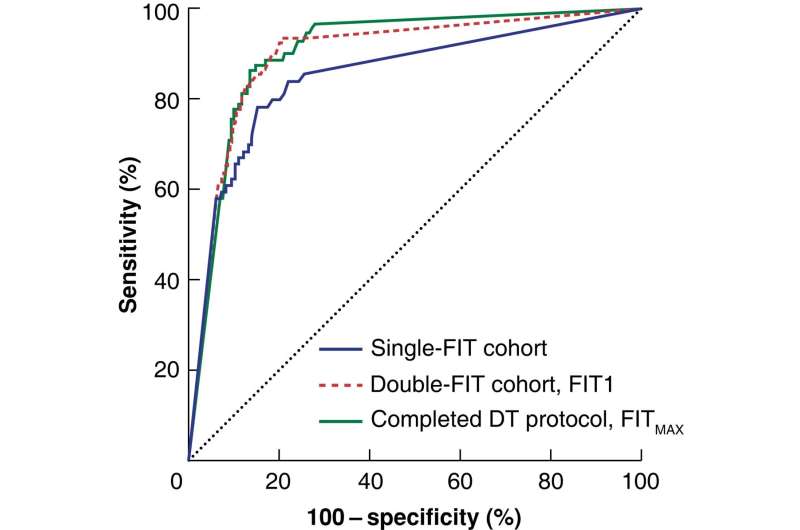
The accuracy of detecting bowel cancer is increased to almost 100% by carrying out a common test twice rather than once, a new study published in the British Journal of Surgery shows.
The test—called a fecal immunochemical test (FIT)—is used to detect blood in bowel movements that is not visible, which may indicate the presence of bowel cancer.
Researchers found that carrying out the test twice ensured more cases of bowel cancer were identified.
As well as reducing the risk of a missed diagnosis, experts say this two-test approach has the potential to reduce demand for colonoscopies in a health care system under pressure.
Early detection
Bowel cancer is the fourth most common cancer in the U.K. and the second biggest cancer killer. More than 16,500 people die from the disease every year.
Early detection is important as bowel cancer is treatable and curable if diagnosed early. Many people have symptoms of bowel disease but none of them are specific for bowel cancer, which can lead to invasive investigations they may not need.
The existing one-test approach is used to determine who should be referred to a specialist, and then once there, to prioritize further investigation for patients whose symptoms suggest they are at a high risk of bowel cancer.
Given the increasing use of FIT as the first investigation for suspected bowel cancer across the U.K., experts say it is important to determine the best use of this test to minimize missing patients with bowel cancer.
Repeated testing
Scientists from the University of Edinburgh used two groups of NHS Lothian patients who had been referred urgently to the Edinburgh Colorectal Surgery Unit to compare the use of one FIT test or two.
They found that doing two FIT tests detected 96.6% of bowel cancer cases correctly, whereas undertaking just one test only detected 84.1%. The median time between the two tests was 13 days.
The study also showed that 16.8% of those to complete two tests had sufficient variation in their test results to change their management plan. This occurred irrespective of significant bowel conditions and highlights the benefit of repeated testing, experts say.
The study showed that patients with two negative FIT tests have a very low underlying risk of bowel cancer (0.17%) and may not need to undergo tests that may cause harm.
Researchers say that while there is an increase in positivity rate by 7.3% using the double-FIT strategy, the workload implications are low given patients with two negative tests may be given alternative management plans.
"Our study highlights how to maximize use of FIT in symptomatic patients. Use of two FIT tests will miss less bowel cancer with a positive impact on patient care. Locally, implementation of the double-FIT strategy has led to a reduction in endoscopy without adversely affecting cancer detection rates," says Farhat Din.
More information: A D Gerrard et al, Double faecal immunochemical testing in patients with symptoms suspicious of colorectal cancer, British Journal of Surgery (2023). DOI: 10.1093/bjs/znad016