This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study shows new chemical compound demonstrates potential in nerve regeneration
Research led by UCL, in partnership with the MRC Laboratory of Molecular Biology (MRC LMB) and AstraZeneca, has identified a new compound that can stimulate nerve regeneration after injury, as well as protect cardiac tissue from the sort of damage seen following a heart attack.
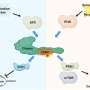
The study, published in Nature, identified a chemical compound, named "1938," that activates the PI3K signaling pathway, and is involved in cell growth. Results from this early research showed the compound increased neuron growth in nerve cells, and in animal models, it reduced heart tissue damage after major trauma and regenerated lost motor function in a model of nerve injury.
Though further research is needed to translate these findings into the clinic, 1938 is one of just a few compounds in development that can promote nerve regeneration, for which there are currently no approved medicines.
Phosphoinositide 3-kinase (PI3K) is a type of enzyme that helps to control cell growth. It is active in various situations, such as initiating wound healing, but its functions can also be hijacked by cancer cells to allow them to proliferate. As a result, cancer drugs have been developed that inhibit PI3K to restrict tumor growth. But the clinical potential of activating the PI3K pathway remains underexplored.
Dr. Roger Williams, a senior author of the study from the MRC Laboratory of Molecular Biology, said, "Kinases are molecular machines that are key to controlling the activities of our cells, and they are targets for a wide range of drugs. Our aim was to find activators of one of these molecular machines, with the goal of making the machine work better. We found that we can directly activate a kinase with a small molecule to achieve therapeutic benefits in protecting hearts from injury and stimulating neural regeneration in animal studies."
In this study, researchers from UCL and MRC LMB worked with researchers from AstraZeneca to screen thousands of molecules from its chemical compound library to create one that could activate the PI3K signaling pathway. They found that the compound named 1938 was able to activate PI3K reliably and its biological effect were assessed through experiments on cardiac tissue and nerve cells.
Researchers at UCL's Hatter Cardiovascular Institute found that administering 1938 during the first 15 minutes of blood flow restoration following a heart attack provided substantial tissue protection in a preclinical model. Ordinarily, areas of dead tissue form when blood flow is restored that can lead to heart problems later in life.
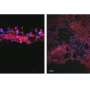
When 1938 was added to lab-grown nerve cells, neuron growth was significantly increased. A rat model with a sciatic nerve injury was also tested, with delivery of 1938 to the injured nerve resulting in increased recovery in the hind leg muscle, indicative of nerve regeneration.
Professor James Phillips (UCL School of Pharmacy), a senior author of the study, said, "There are currently no approved medicines to regenerate nerves, which can be damaged as a result of injury or disease, so there's a huge unmet need. Our results show that there's potential for drugs that activate PI3K to accelerate nerve regeneration and, crucially, localized delivery methods could avoid issues with off-target effects that have seen other compounds fail."
Given the positive findings, the group is now working to develop new therapies for peripheral nerve damage, such as those sustained in serious hand and arm injuries. They are also exploring whether PI3K activators could be used to help treat damage in the central nervous system, for example due to spinal cord injury, stroke or neurodegenerative disease.
Professor Bart Vanhaesebroeck (UCL Cancer Institute), a senior author of the study, said, "This is a prime example of interdisciplinary research, in which people with expertise ranging from basic science, drug development and clinical studies join forces around an innovative idea, while also crossing boundaries between academia and industry."
"Blue sky research of this kind is difficult to get funding for in a world of increasing specialization, but hopefully this project can provide something of a model for future ambitious research."
More information: Bart Vanhaesebroeck, A small-molecule PI3Kα activator for cardioprotection and neuroregeneration, Nature (2023). DOI: 10.1038/s41586-023-05972-2. www.nature.com/articles/s41586-023-05972-2