This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Team reports on development of potential new generic treatment for multiple types of cancer
Researchers led by Katsunori Tanaka at the RIKEN Cluster for Pioneering Research (CPR) in Japan and Hiromitsu Haba at the RIKEN Nishina Center for Accelerator-Based Science (RNC) have developed a new technique that has the potential to generically treat several kinds of cancer, with fewer negative side effects than currently available methods.
Published on June 27 in Chemical Science, the proof-of-concept study showed that tumors in mice grew almost three times less and survival was 100% after just one injection of a compound that is designed to emit small amounts of alpha radiation from the inside of cancer cells, thus killing them but sparing healthy tissue.
The side effects of standard chemotherapy and radiation treatment can be devastating, and the eradication of all cancer cells is not guaranteed, especially when the cancer has already metastasized and spread throughout the body. Therefore, the goal of most research these days is to find a way to specifically target cancer cells so that treatments only affect tumors. Some targeted treatments do exist, but they cannot be applied to all cancers.
"One of the greatest advantages of our new method," says Tanaka, "is that is can be used to treat many kinds of cancer without any targeting vectors, such as antibodies or peptides."
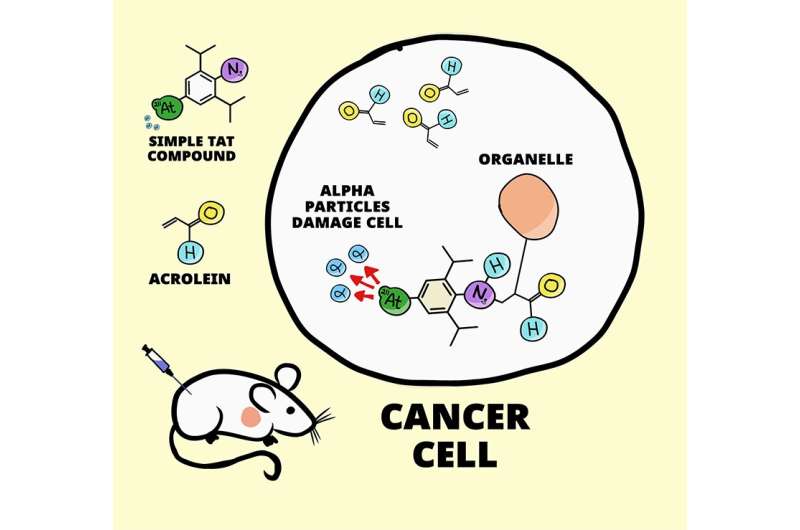
The new technique relies on basic chemistry and the fact that a compound called acrolein accumulates in cancer cells. A few years ago, Tanaka's team used a similar technique to detect individual breast cancer cells. They attached a fluorescent compound to a specific type of azide—an organic molecule with a group of three nitrogen atoms (N3) at the end.
When the azide and acrolein meet inside a cancer cell, they react, and the fluorescent compound becomes anchored to structures inside the cancer cell. Because acrolein is almost absent from healthy cells, this technique acted like a probe to light up cancer cells in the body.
In the new study, rather than simply detecting cancer cells, the team targeted those cells for destruction. The logic was fairly simple. Instead of attaching the azide to a fluorescent compound, they attached it to something that can kill a cell without harming surrounding cells. The chose to work with astatine-211, a radionuclide that emits a small amount of radiation in the form of an alpha particle as it decays.
Compared to other forms of radiation therapy, alpha particles are a little more deadly, but they can only travel about one-twentieth of a millimeter and can be stopped by a piece of paper. In theory, when astatine-211 is anchored to the inside a cancer cell, the emitted alpha particles should damage the cancer cell, but not much beyond.
Once the team figured out the best way to attach astatine-211 to the azide probe, they were able to perform a proof-of-concept experiment to test their theory. They implanted human lung-tumor cells into mice and tested the treatment under three conditions: simply injecting astatine-211 into the tumor, injecting the astatine-211-azide probe into the tumor, and injecting the astatine-211-azide probe into the bloodstream.
They found that without targeting, tumors continued to grow, and mice did not survive. As expected, when the azide probe was used, tumors grew almost three times less and many more mice survived—100% when it was injected into the tumor and 80% when injected into the blood.
"We found that just one tumor injection with only 70 kBq of radioactivity was extremely effective at targeting and eliminating tumor cells," says Tanaka. "Even when injecting the treatment compound into the bloodstream, we were able to achieve similar results. This means we can use this method to treat very early-stage cancer even if we don't know where the tumor is."
The fluorescent probe version of this technique is already being tested in clinical trials as a way of visualizing and diagnosing cancer at the cellular level. The next step is to find a partner and begin clinical trials using this new method to treat cancer in humans.
More information: Ode et al, Therapeutic efficacy of 211At-radiolabeled 2,6-diisopropylphenyl azide in mouse models of human lung cancer, Chemical Science (2023). DOI: 10.1039/d3sc02513f