This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers identify new treatment targets for chronic pain patients
Exposure to opioids under chronic pain conditions produces unique gene expression changes in brain regions associated with cognition, emotion, and reward, according to preclinical research conducted at the Icahn School of Medicine at Mount Sinai and published June 8 in Nature Neuroscience. The study highlights new molecules that might serve as specific targets for chronic pain patients to enable them to move beyond opioid treatments.
Chronic pain and opioid use disorder are two highly prevalent conditions. During the opioid epidemic in the United States, many chronic pain patients became reliant on readily accessible drugs such as oxycodone. Recent efforts to reduce oxycodone maintenance in these patients have led many to undergo opioid withdrawal even as they still experience chronic pain. Therefore, the identification of non-opioid medications that alleviate pain while facilitating withdrawal from opioids would be a major advance in the field.
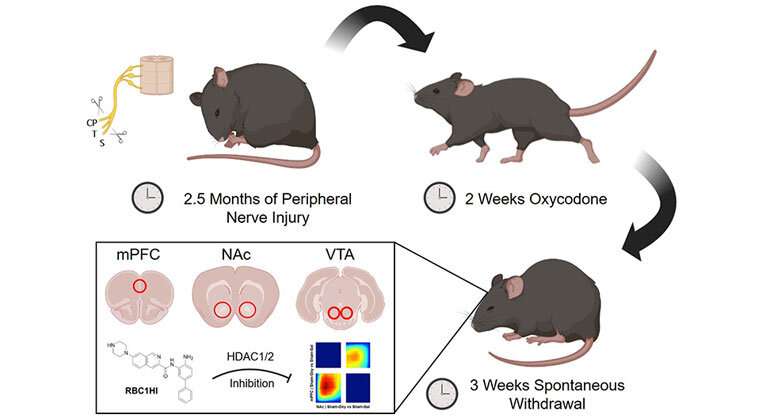
To better understand the effects of chronic opioid exposure and physical dependence on the brain reward circuitry of those with chronic pain, the research team administered high doses of oxycodone for two weeks to mice with nerve injury and a group of control mice. Following three weeks of spontaneous withdrawal, they collected tissue from the medial prefrontal cortex (mPFC), the nucleus accumbens (NAc), and the vental tegmental area (VTA).
The team used RNA sequencing, a high-throughput method that measures messenger RNA—molecules that allow DNA to change the amount of a specific protein within a cell in response to environmental factors, like a nerve injury or prolonged drug exposure.
"We identified changes in the expression of hundreds of genes within the brain's reward circuitry associated with oxycodone physical dependence," said Venetia Zachariou, Ph.D., Adjunct Professor of Neuroscience at Icahn Mount Sinai; Professor and Chair of Pharmacology, Physiology, and Biophysics at Boston University Chobanian & Avedisian School of Medicine; and senior author of the paper, who led the work at Mount Sinai.
"Our data revealed that chronic neuropathic pain states affect sensory, affective, and molecular signatures of opioid withdrawal."
The investigators applied bioinformatics analyses to predict changes in the activity of transcription factors—proteins that control gene expression—under different pain/opioid withdrawal states. One of the identified targets was a protein called histone deacetylase 1 (HDAC1). This protein regulates gene transcription by reducing the accessibility of the transcriptional machinery to certain genes that might help cells overcome injury.
Dr. Zachariou's team was also able to test a new blood-brain-barrier-permeable HDAC1/2 inhibitor, named RBC1HI, in this new mouse model of opioid dependence under chronic pain. Treatment with RBC1HI prevented the development of opioid withdrawal while simultaneously alleviating the sensory and emotional signs of neuropathic pain.
"The novel animal model used in this study revealed very different effects of opioids in the presence or absence of a pain syndrome," said Eric J. Nestler, MD, Ph.D., the Nash Family Professor of Neuroscience, director of the Friedman Brain Institute, and Dean for Academic Affairs at Icahn Mount Sinai, and chief scientific officer of the Mount Sinai Health System.
"Data from this study will be valuable for identifying and validating several new targets like HDAC1 across chronic pain- and opioid withdrawal-associated conditions. These targets in turn offer new avenues toward developing non-opioid treatments of chronic pain and may also be helpful for the development of treatments for substance use disorders."
More information: Kerri D. Pryce et al, Oxycodone withdrawal induces HDAC1/HDAC2-dependent transcriptional maladaptations in the reward pathway in a mouse model of peripheral nerve injury, Nature Neuroscience (2023). DOI: 10.1038/s41593-023-01350-3