This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
BUB1 regulates EGFR signaling by reducing EGFR internalization
A new research paper titled "Budding uninhibited by benzimidazoles-1 (BUB1) regulates EGFR signaling by reducing EGFR internalization" has been published inAging.
EGFR signaling initiates upon ligand binding, which leads to activation and internalization of the receptor-ligand complex. In this new study, researchers Shyam Nyati, Grant Young, Corey Speers, Mukesh K. Nyati, and Alnawaz Rehemtulla from the University of Michigan, Henry Ford Health System and Case Western Reserve University evaluated whether BUB1 impacted EGFR signaling by regulating EGFR receptor internalization and activation.
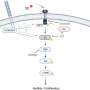
"We postulate that BUB1 helps in the formation and stabilization of EGFR dimers at the membrane and may regulate endocytosis of activated EGFR into either clathrin dependent (EEA1 coated) or independent (caveolin coated) vesicles thus impacting receptor recycling or degradation and subsequently signaling amplitude and duration," the researchers explain.
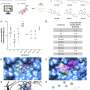
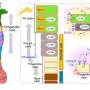
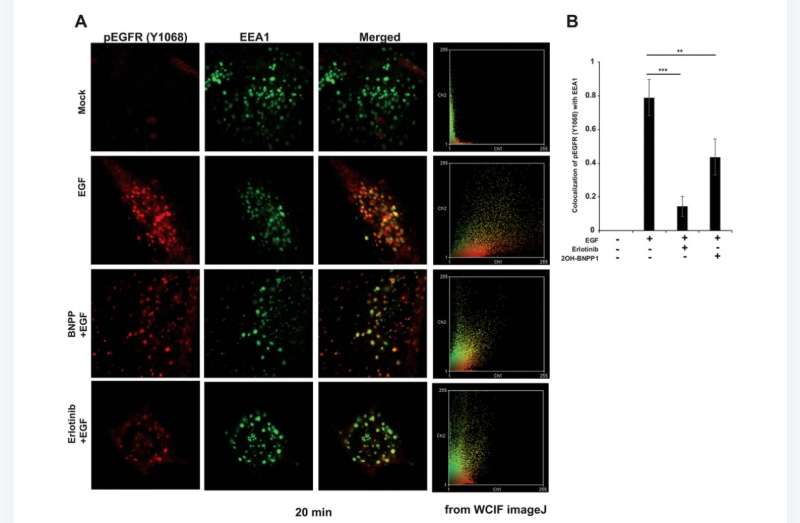
Within the research, BUB1 was ablated genomically (siRNA) or biochemically (2OH-BNPP1) in cells. EGF ligand was used to initiate EGFR signaling while disuccinimidyl suberate (DSS) was used for cross linking cellular proteins. EGFR signaling was measured by western immunoblotting and receptor internalization was evaluated by fluorescent microscopy (pEGFR (pY1068) colocalization with early endosome marker EEA1).
The research team found that siRNA-mediated BUB1 depletion led to an overall increase in total EGFR levels and more phospho-EGFR (Y845, Y1092, and Y1173) dimers while the amount of total EGFR (non-phospho) dimers remained unchanged. BUB1 inhibitor (BUB1i) decreased EGF mediated EGFR signaling including pEGFR Y845, pAKT S473 and pERK1/2 in a time-dependent manner. Additionally, BUB1i also reduced EGF mediated pEGFR (Y845) dimers (asymmetric dimers) without affecting total EGFR dimers (symmetric dimers), indicating that dimerization of inactive EGFR is not affected by BUB1.
Furthermore, BUB1i blocked EGF mediated EGFR degradation (increase in EGFR half-life) without impacting half-lives of HER2 or c-MET. BUB1i also reduced co-localization of pEGFR with EEA1 positive endosomes suggesting that BUB1 might modulate EGFR endocytosis.
The researchers conclude, "Our data provide evidence that BUB1 protein and its kinase activity may regulate EGFR activation, endocytosis, degradation, and downstream signaling without affecting other members of the receptor tyrosine kinase family."
More information: Shyam Nyati et al, Budding uninhibited by benzimidazoles-1 (BUB1) regulates EGFR signaling by reducing EGFR internalization, Aging (2023). DOI: 10.18632/aging.204820