This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
preprint
trusted source
proofread
Human vs machine: A closer look at nerve insulating cells could be a boon for brain-computer interface, and much more
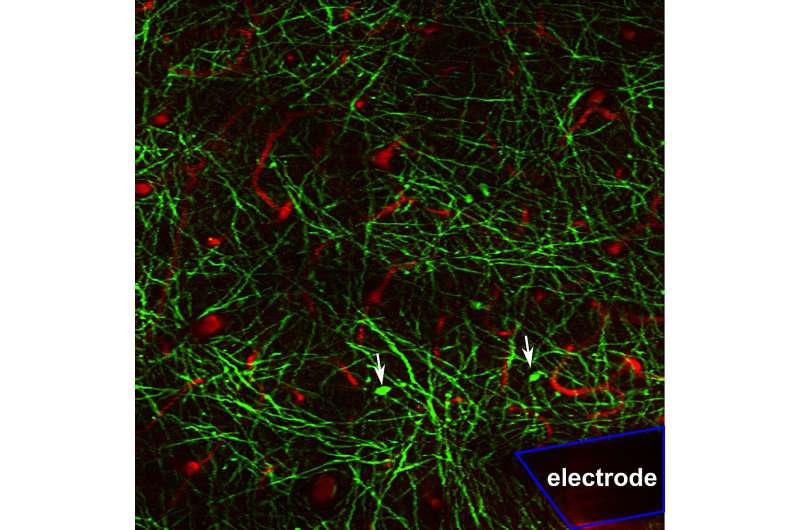
Brain-computer interface (BCI)—an experimental technology that makes it possible to move robotic limbs using only one's thoughts—holds promise to transform the lives of people with paralysis. But before BCI can make the leap from the lab to patients' everyday lives, a nagging problem remains: how to coax the body to coexist, for the long haul, with the implants.
"There are two major challenges," says Takashi (TK) Kozai, associate professor of bioengineering at the University of Pittsburgh Swanson School of Engineering, who leads the BIONIC Lab (Bio-Integrating Optoelectric Neural Interface Cybernetics). For one, over time, scar tissue forms around the electrodes, which complicates the signal's path to the nearest neuron and dampens the voltage. And for another, there's a lot of variability in performance.
"Sometimes these devices work, and sometimes they don't," he says.
Overcoming these challenges will be a formidable feat from both the engineering and neurobiology perspectives. Luckily, Kozai lives right at the nexus of the two.
The focus of his project is to gain a better understanding of a specific cell type, called oligodendrocytes, which he believes could play a key role in settling this unique human-versus-machine dilemma.
Oligodendrocytes form the insulative coating known as myelin, which makes neural connections in the brain much faster and more streamlined. Without this insulation, what's left is a long line of naked axons straining to send impulses along jagged, roundabout paths. That inefficiency, Kozai realized, doesn't just cause a slowdown, but also means these neurons have to work much harder than their myelinated counterparts, wasting precious energy. "So oligodendrocytes are essentially energy savers," he says.
If Kozai's hunch is correct about oligodendrocytes—an historically understudied cell in his research specialty—this work could have broad implications for a number of debilitating conditions.
Historically, the thinking has been that the trouble with the BCI starts like this: As a result of electrode implantation, blood vessels are damaged, which sends plasma proteins flying off where they shouldn't be. Their presence on the wrong side of the blood-brain barrier unleashes an assault of immune cells, called microglia. And in that fallout, neurons die.
But the team began observing that the friendly fire wasn't the only problem; it was that these neurons, having been choked off from their blood supply, had simply starved to death. So Kozai wondered if they could somehow fortify the neurons against that starvation. "That's what pointed us toward the oligodendrocytes."
With this new R01 grant, Kozai is targeting certain progenitor cells that each have potential to give rise to either an oligodendrocyte or enemy number one: scar tissue. In a preliminary study, the team treated these cells with a drug to encourage the former. When administered before implant surgery, they found, the drug helped more neurons survive after the procedure.
The project will also probe oligodendrocyte health more generally, insights that could potentially expand our understanding of traumatic brain injury and stroke—which both involve a decrease in blood flow in the brain—as well as multiple sclerosis (MS)—an autoimmune onslaught on neurons that's initially sparked by oligodendrocyte death.
Kozai's group may be one of the first to have suggested that preserving oligodendrocytes could be a novel treatment against dementia in Alzheimer's. In that disease, the focus has long been on the plaques that form in the brain.
"But there's growing evidence that it's actually the blood vessels that suffer first—the plaques form on the blood vessels before they form on the neurons," Kozai explains. Amid the ensuing blood loss, it's possible the neurons are more vulnerable, unable to maintain a clean environment, "and that, in turn, leads to the plaque forming on the neurons."
The findings are available on the bioRxiv preprint server.
More information: Keying Chen et al, Pro-myelinating Clemastine administration improves recording performance of chronically implanted microelectrodes and nearby neuronal health, bioRxiv (2023). DOI: 10.1101/2023.01.31.526463