This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists unravel the mechanisms behind a key culprit in chemotherapy resistance
Most new cancer cases are treated with chemotherapy, but humans naturally express proteins that indiscriminately remove chemotherapy drugs from many cells—including tumor cells, which reduces treatment efficacy.
New research out of St. Jude, along with Australian and English collaborators, has discovered how one of these cellular-cleaning proteins, ABCG2, removes many kinds of chemotherapeutics from a cell and how to prevent it, which could improve future anti-cancer therapies. The findings were published August 18 in Nature Communications.
Chemotherapy involves a course of one or several drugs that usually take the form of small molecules. However, our body employs a family of proteins called ATP-binding cassettes (ABC) to function as cellular cleaners, responsible for collecting and transporting excess chemicals from the cell.
While their cause is a noble one, they often cause a significant issue in chemotherapy—the ABCs, particularly the protein ABCG2, do too good a job of cleaning up the cell and removing the administered drugs, often before they can affect the tumor.
"ABCG2 plays a role in clearing toxins and protecting healthy stem cells from chemotherapy and toxic chemotherapeutic agents," said John Schuetz, Ph.D., St. Jude Department of Pharmacy and Pharmaceutical Sciences. "We showed a few years ago that ABCG2 levels were quite high in certain subtypes of medulloblastoma, which impacted how much of the drug could get in to kill the cancer cells," Schuetz said. "In this study, we found why the protein is able to clear out such a broad spectrum of chemo drugs, which can now inform efforts to improve anti-cancer strategies."
Water-loving amino acids hint at mechanism
The problem is that ABCG2 excels at its job. This can be disastrous to therapeutic endeavors. No matter how a drug is designed, ABCG2 has the tools to remove it from the cell. Therefore, Schuetz and his colleagues take an interrogative approach to answer how ABCG2 removes chemotherapies from cells by probing the substrate binding site, looking for its tools.
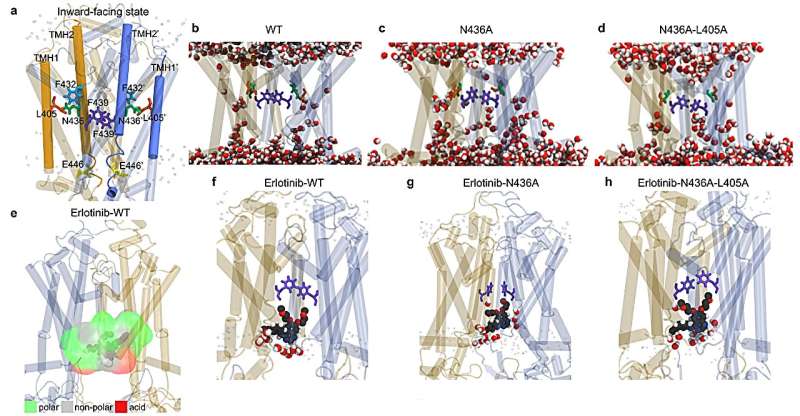
"We were completely puzzled by ABCG2's promiscuity until we did these studies," Schuetz said. "We found that unlike some of its genetic family members, which have a very hydrophobic binding pocket, the ABCG2 pocket is dotted with hydrophilic residues as well as hydrophobic ones."
ABCs are usually responsible for removing hydrophobic (water-hating) molecules, such as lipids. Other transporters take care of hydrophilic (water-loving) molecules. ABCG2 is unique because it is involved in removing both, making it capable of removing many types of anti-cancer drugs from cells and limiting therapeutic efficacy. In effect, it has more removal tools than its family members.
The researchers set out to find the exact residues giving ABCG2 the capability to bind and remove hydrophilic molecules. Two amino acids stand out within the substrate binding site of ABCG2—a threonine and an asparagine. The distinguishing feature of these amino acids is that they are polar and therefore hydrophilic. To test if these two residues were indeed the culprits for ABCG2's promiscuity, the researchers turned each amino acid in the substrate binding site one by one into a featureless alanine and observed how each mutation affected the transport of small molecules into cells.
Locking the door on hydrophilics
The researchers expected to find a gradual decrease of ABCG2's ability to remove all its target molecules from the cell. Instead, they discovered that while most of the mutations did result in a moderately decreased ability to bind substrates and transport them across the membrane, thus removing them from the cell, some capabilities were actually increased. For hydrophobic molecules, the ABCG2 with an alanine instead of an asparagine was better at transporting these compounds—two-fold better. Meanwhile, its ability to transport hydrophilic compounds was almost completely removed.
"The protein toggles like a one-way door to be open on one side of the membrane or the other to move things out of the cell. This movement excludes any water," Schuetz explained. "With this asparagine to alanine mutation, it's allowing some water to move in. This moves the hydrophilic substrates and locks them in tight, so they stick around much longer and are not transported very well."
Potential paths to combat drug resistance
The work highlights a way forward to combat drug resistance, especially in cancer. ABCG2 inhibitors are often combined with chemotherapies, but preventing ABCG2 function can result in off-target detrimental effects. "The goal is to design ABCG2 inhibitors that have minimal effect on normal tissues but target the tumor," Schuetz said.
With the knowledge gained from this work, more effective, less detrimental inhibitors can be designed to target the binding site threonine in ABCG2 when paired in combination with hydrophilic molecules used in chemotherapy.
More information: Tomoka Gose et al, The net electrostatic potential and hydration of ABCG2 affect substrate transport, Nature Communications (2023). DOI: 10.1038/s41467-023-40610-5