This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study opens new frontier for research into neurodevelopmental disorder
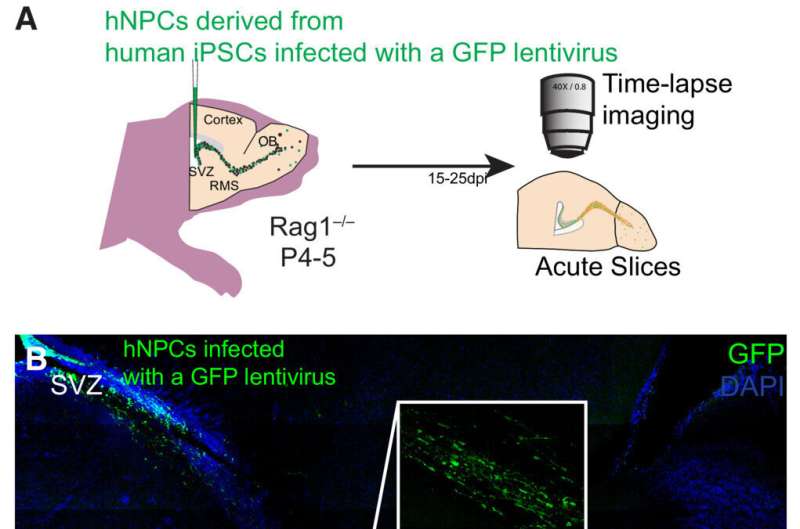
An international team led by a newly appointed Canada Research Chair at the University of Ottawa's Faculty of Medicine has shed light on the underlying mechanisms of a mysterious brain abnormality that occurs during human fetal development.
By uncovering these hidden mechanisms, scientists could potentially design new strategies to better understand "Periventricular heterotopia" (PH) and prompt bench-to-bedside translation from the laboratory into the clinic to efficiently treat this devastating disorder.
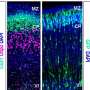
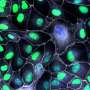
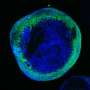
The collaborative research team led by Dr. Armen Saghatelyan aimed to uncover the migratory mechanisms of grafted human neuronal progenitor cells derived from PH patients in the brains of an immunodeficient mouse model. They wanted to test the hypothesis that changes in autophagy—a kind of cellular recycling process in which a cell turns on itself to discard unwanted material—impact the neuronal cells' abnormal migratory behaviors.
PH is a neurodevelopmental affliction that's characterized by an abnormal migration of neuronal cells. These cells end up clustering around ventricles, the cavities of the brain. The disorder typically becomes apparent with recurrent seizures. It is genetically heterogenous, meaning that mutations in distinct genes may lead to PH.
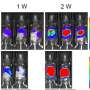
With rigorous experiments using the FDA-approved drug metformin to modulate autophagy, the team found the drug restored the cells' migratory properties by triggering autophagy.
"Since metformin is already used for the treatment of diabetes and in pediatric clinical trials, our results open up the possibility of pharmacological interventions in PH patients to counteract, at least partially, the migratory deficits of neuronal cells causing a critical malformation," says Dr. Saghatelyan, who was recently named Canada Research Chair in postnatal neurogenesis and is a professor in the Department of Cellular and Molecular Medicine.
"We hope that our findings may pave a way for pharmacological intervention in PH patients to counteract the migratory deficits of neuronal cells induced by PH-inducing genetic mutations."
Metformin is a commonly prescribed drug. The oral medication was introduced decades ago and is a first-line treatment for Type 2 diabetes, helping people improve their blood-sugar control and sensitivity to insulin.
Prior to this study, the underlying mechanisms of PH neuronal cell migration was unknown. It was also totally unclear whether migratory deficits might be restored by modulating other pathways not affected by the genetic mutations.
Dr. Saghatelyan served as the study's senior author. Cedric Bressan, a Ph.D. student in his lab, is the first author. Dr. Saghatelyan says the core component of the research was the development of strategies to transplant human neuronal cells into the brains of immunodeficient mice and using various live imaging approaches to monitor their dynamics and migratory characteristics. This work started eight years ago, first in his lab at Université Laval, and later at his lab at uOttawa.
The work is published in the journal EMBO Molecular Medicine.
More information: Cedric Bressan et al, Metformin rescues migratory deficits of cells derived from patients with periventricular heterotopia, EMBO Molecular Medicine (2023). DOI: 10.15252/emmm.202216908