This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Researcher inhibits flu viruses by blocking the enzyme that cleaves off virus particles

A flu virus could cause a pandemic, and we would be poorly armed because flu viruses are starting to become resistant to flu medications like Tamiflu. Chemist Merijn Vriends successfully worked on an improved version of such medications. He will be awarded his doctorate on September 12.
School closures and other drastic measures seemed unreal and distant when Merijn Vriends began his research in 2019 and read about the consequences of a pandemic. "We have all experienced it now." It could all happen again, even due to a flu virus. "For example, if bird flu becomes transmissible among humans or if other types of flu viruses mutate. During the last flu pandemic in 2009, the consequences were relatively mild, but it could have been different."
Resistant to Tamiflu
In his research, Vriends focused on inhibiting flu viruses. "The most commonly used agents for that purpose are Tamiflu and Relenza, but some types of flu are resistant or can become resistant. I developed molecules that work through a different mechanism than these drugs."
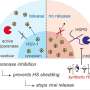
Virus particles need their host to multiply. A flu virus enters one of our cells and activates our machinery to create new virus particles. These particles leave the cell and infect new cells and/or new victims. Upon leaving the cell, the offspring is still attached to sialic acid: a sugar molecule on the outside of our cells. Then, an enzyme from the neuraminidase family cleaves the virus. Existing virus inhibitors like Tamiflu block neuraminidase, preventing it from cleaving the virus particles. The problem is that this blockade is reversible.
A new segment blocks the enzyme irreversibly
Vriends states, "I succeeded in complementing an existing inhibitor for flu viruses with a molecular segment that strongly binds to the cleaving enzyme neuraminidase and irreversibly blocks it." With the new piece attached to it, the enzyme can no longer cleave sialic acid. Newly formed virus particles remain attached to the cell in which they were formed and cannot spread further.
The Ph.D. candidate conducted many experiments in the lab. "I started with a starting material and modified it with various types of reactions, always moving towards the desired molecule. Step by step, you build toward the final molecule. The molecules turned out to be very difficult to make. Many reactions I tried did not work or gave a low yield. So it became a matter of trying a lot and often going back to the drawing board."
A research group in Barcelona conducted calculations on the connections Vriends was trying to make. "This way, we examined what spatial shape the new molecule would assume and how it interacts precisely with the enzyme."
It was not at all sure that the project would succeed, according to Vriends. "It was very satisfying to actually have the molecule I wanted after trying for a very long time. Then we saw that it really does what we want. That is not always easy to control."
Virus inhibitors with this new molecular group attached are not expected to be available at the pharmacy for the time being. "They are still difficult to produce on a large scale, and we do not yet know if they are safe for use in humans or animals."
However, the molecules from Vriends' thesis are already handy tools for researchers. "You can study neuraminidase enzymes with them. We are already doing that with a research group in York. If we better understand these enzymes, it can contribute to the timely control of new, dangerous flu types," says Vriends.