This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
reputable news agency
proofread
With diabetes on the rise, tech becomes crucial part of disease management
At the Hennepin Healthcare Center of Diabetes and Endocrinology in downtown Minneapolis, 66-year-old Mark Harris tries to recall his medication intake and sleep schedule from the past week.
Laura LaFave, an endocrinologist at the center, listens attentively while seated at a computer desk, looking at metrics of Harris' blood sugar levels extracted from the Bluetooth-connected continuous glucose monitor (CGM) he wears daily. Harris' glucose levels are low, which leads LaFave to pry deeper into how his insulin pump is functioning.
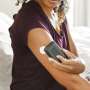
At the center, as many as 70% of patients with diabetes wear some sort of glucose monitoring, insulin-administering technology, LaFave said. It's vital the device works properly for Harris, a Type 1 diabetic, meaning his body produces little to no insulin. The combination of an insulin pump and CGM allows the pump to adjust insulin automatically injected into his abdomen through a slender plastic tube. The CGM tests his glucose levels through sensors patched to his skin.
For the roughly 500,000 Minnesotans diagnosed with diabetes, technology that automates the formerly manual finger-pricking and shot-giving has been an essential development and a relief for many who now can trust technology instead of just themselves to help manage a disease that was the seventh-leading cause of death in the U.S. in 2019, according to the American Diabetes Association.
Nationwide, more than 10 million Americans diagnosed with diabetes use insulin and benefit from CGM devices, per the association. That's out of 133 million currently living with diabetes or prediabetes in the U.S.
"The technology is new, and we are still learning how all the pieces fit together, but there's no question that we're starting to see treatments and technology that can transform the lives of people with diabetes," said Francisco Prieto, chair of the National Advocacy Committee within the American Diabetes Association.
A tech solution
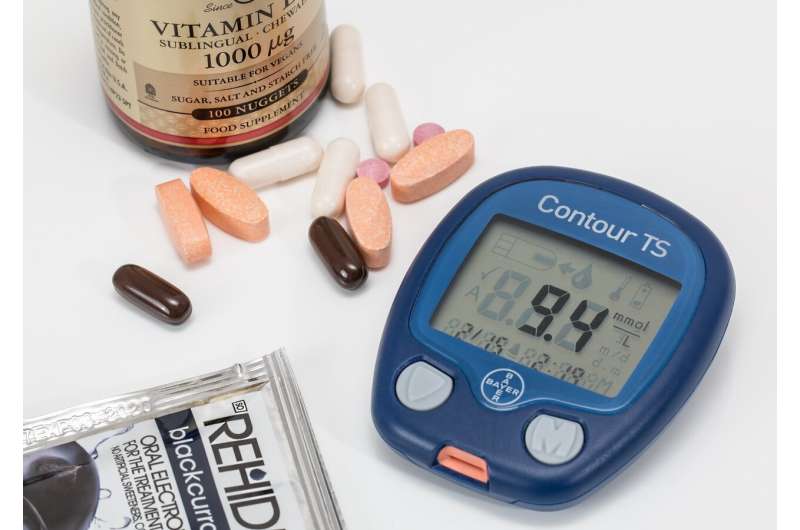
For so long, the burden was on the patient to know their medication doses and glucose levels, said LaFave, who's worked with diabetics for 16 years. The finger prick remains a barrier, as some are reluctant to do it.
With technology, the ability to remotely download a patient's data from a CGM and conduct follow-up consultations has changed the delivery of care, LaFave said.
"There's good studies showing that when patients are able to see that data laid out, and they can go through what happened in this period of time, that really increases engagement of their care," she said.
That's the premise of CGM innovation at companies like Abbott, said Jared Watkin, senior vice president of the Illinois-based medical technology company's diabetes care business. Abbott is one of four major providers of CGMs along with Medtronic, whose operational headquarters are in Fridley; Dexcom; and Senseonics.
Watkin worked on the research and development team that created the company's first generation FreeStyle Libre CGM, first launched in Europe in 2014. Now on its third version, more than 5 million users wear it worldwide, including 2 million in the U.S. A companion smartphone app allows friends, family and caregivers to follow care remotely.
Abbott recently won FDA breakthrough designation for a new sensor that would measure glucose and ketones at the same time, Watkin said. Ketones are acidic and can cause major disruptions to the body's biochemistry.
Before having a CGM, Harris was in the intensive care unit for ketoacidosis, a condition that develops when the body can't transfer glucose into cells, and broken-down fat creates ketones. He was homeless at the time, and insulin was hard to find.
A year after that in 2010, he received his first CGM through a medical-assistance program.
"We think this can be a very powerful new tool, certainly in Type 1 diabetes management in children and adults, because it tells you when you need to do something about it and need to contact a health care professional," Watkin said. "In the past, they wouldn't have necessarily been doing that."
Adding other measurables to sensors, not just a single sensor for glucose in Type 1 diabetics, could offer more benefits to people, Watkin said. That includes sensors for people who might be prediabetic or not yet in full Type 2 diabetes who can use readings from sensors for pharmaceutical or diet-based prevention.
More innovation needed
A Minneapolis tech company is working with the Centers for Disease Control and Prevention (CDC) to engage millions of prediabetic people.
In September, the CDC awarded AmVentureX a $12.5 million grant to help scale the health organization's national diabetes prevention program. AmVentureX's app, called BioCoach, focuses on metabolic lifestyle management through subscription-based content and curated meals shipped to users' homes.
The company also makes and sells a variety of take-home test kits and devices, including a ketone monitor that users can connect to the app for better health management.
AmVentureX will use the funding through the next five years to market the CDC's program and partner with community organizations, insurance companies and health systems to reach people at risk of developing Type 2 diabetes. The funding also helps AmVentureX drive people to the BioCoach app, Chief Executive Matt Payne said.
Through the app, users upload and monitor their metabolic health information. A multilingual team of health experts provide meal suggestions, fitness tips and other daily routines based on a person's health data, Payne said. The cost for a subscription is $35 per month.
By the end of 2024, Payne expects to have as many as 30,000 users on the app, more than double what it has now.
Though medical device companies developed CGMs for Type 1 diabetics, and thus those are the majority of users, 95% of people with diabetes are Type 2, meaning they are insulin resistant.
With research, more tech-focused solutions are in the pipeline for those with Type 2, Watkin said.
"The great news is we've got 5 million people using FreeStyle Libre, and a fair number of them around the world now have Type 2 diabetes, and we're able to analyze that data, and it shows that these sensors are really just as effective in Type 2 diabetes as they are in Type 1," he said. "Those lessons of ease of use, affordability and accessibility, you've got to put that on steroids for the Type 2 market because it's such a large group of people."
The use of artificial intelligence would accelerate the ability to deliver care through CGMs, Watkin said. AI, Watkin said, introduces an opportunity to determine personalized care using not only data on glucose levels but a person's overall health picture, where AI systems can recommend insulin doses, exercises and evening meals to keep blood sugar at certain levels.
A new ally
Harris received his Type 1 diagnosis more than 40 years ago. In the 1970s, glucose test strips that required urine or blood samples were his only options, along with having to inject himself with insulin.
With his previous CGM, Harris had to manually input glucose readings. And while Harris still pricks his finger—just to double check his CGM—that practice could be obsolete in many parts of the world within a decade, Watkin said.
"There are children diagnosed with diabetes who never pricked their finger," Watkin said. "... Would I say they're going to disappear across the world? Probably not because there's going to be certain barriers. But from large parts of the population around the world, they will not be putting any children on blood testing. It will be CGM as a standard care."
Harris lives alone, so having technology to manage his illness enables him to live a somewhat normal life with less fear of having a diabetic episode.
"I don't have to rely on myself," he said, "if I'm having low blood sugar."
©2023 StarTribune.
Distributed by Tribune Content Agency, LLC.