December 7, 2023 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study shows that AgRP neurons encode circadian feeding time in mice
Animals and humans follow specific daily patterns known as circadian rhythms, which naturally prompt their bodies to sleep, wake up, eat and have other physiological responses at specific times. The disruption of these rhythms can have adverse effects on both their health and psychological well-being.
Neuroscientists have been trying to understand the neural underpinnings of circadian rhythms for decades, as this could help to devise more effective treatments for mental health disorders characterized by the disruption of normal sleeping or eating patterns.
A paper by researchers at University of Iowa and Yeditepe University, published in Nature Neuroscience, identified a class of neurons that could encode the times at which mice, and potentially other mammals, regularly eat.
"We used to think that hunger slowly creeps in as available energy resources diminish over time," Deniz Atasoy, one of the researchers who carried out the study, told Medical Xpress.
"Since AgRP neurons are one of the master regulators of hunger responses in the brain, we expected them to show an activity pattern like this, i.e. slowly increased firing rate as we get hungrier. This is also what you would expect from a classical 'homeostatic' regulation such that any deviation from determined values, but in this case, available nutrients such as glucose would trigger behavioral and physiological changes to counter this to bring these values back to a set point."
The primary objective of the study by Atasoy and his colleagues was to investigate the role of AgRP neurons in regulating feeding time, through a series of experiments carried out on living mice. Many previous studies focusing on hunger-regulating neurons were partly limited and did not paint a clear picture of their function.
"Previous efforts investigating the function of hunger-regulating neurons function were limited to very short time scales like minutes, whereas hunger physiology operates over hours and even days," Atasoy explained.
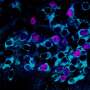
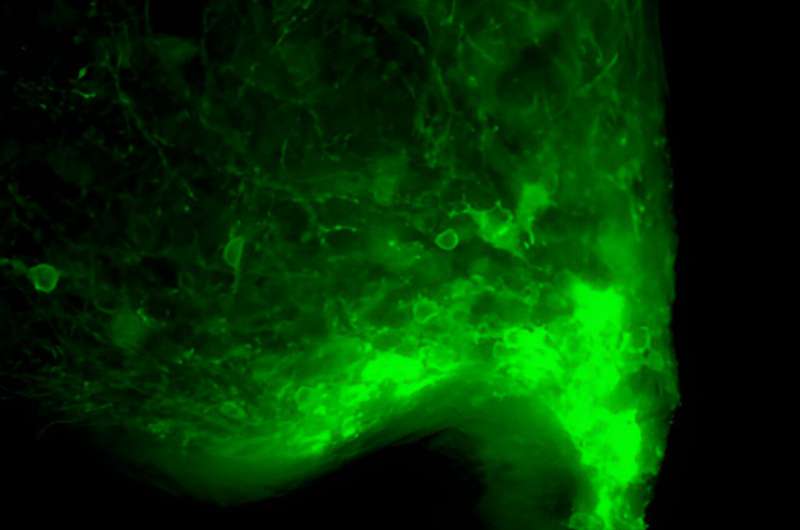
"Monitoring the activity of a defined set of neurons, in this case hypothalamic AgRP neurons, for days is technically challenging, and we managed this using a technique called fiber photometry on mice. This approach is based on making the neuron population of interest literally fluorescent when they become active."
The researchers subsequently placed a tiny probe in proximity to the AgRP neurons that they had made fluorescent using fiber photometry. This allowed them to pick up and monitor their activity, based on the light signals produced when the neurons started firing.

These experiments allowed Atasoy and their colleagues to detect when the mice felt the urge to eat, observing the activity of the AgRP neurons around that time. Contrary to theoretical predictions, their findings suggest that AgRP neurons do not work as a homeostat (i.e., firing when energy is low and ceasing their activity once the mice finish eating).
"Instead, we found that ups and downs in daily AgRP neuron activity appear to follow recent feeding pattern," Atasoy said. "A yet to be clarified biological clock appears to be synchronized to past mealtimes and turns on and off the activity of AgRP neurons at those mealtimes, This is a clear divergence from 'homeostatic' regulation and more in line with so-called 'allostatic' regulation. Unlike homeostasis, in allostatic regulation, animals do not wait until their energy level drop."
The results gathered by Atasoy and their colleagues suggest that AgRP neurons proactively defend the energy levels of mice before they actually feel the need to eat. They seem to achieve this by predicting future energy deficits based on cues learned from the past experiences of mice.
"In this case, recent history of circadian feeding time appears to be the entraining cue," Atasoy said. "Humans and mice share very similar basic circuitry of feeding and satiety, thus we believe these observations are likely to extend humans as well. Consistently, rhythmic nature of hunger has been shown in humans by other groups before, our findings provide mechanistic insight into these observations."
The recent work by this team of researchers gathered new insights about how AgRP neurons contribute to the feeding patterns of mice. If they were validated in humans, these findings could inform medical research, for instance paving the way for the development of new treatments for disorders characterized by dysregulated eating patterns.
"These results opened a whole new window of previously overlooked contribution of the time of day in appetite physiology," Atasoy added.
"In our next studies, we would like to find out what the molecular mechanism of this food-tuned 'clock' is. Knowing how it works might lead to therapeutic technologies for better-informed control over their activity. Another question raised is whether satiety-promoting neurons have such rhythms. If so, how does it affect their activity, responsiveness to satiety signals and even anti-obesity treatments?"
More information: Nilufer Sayar-Atasoy et al, AgRP neurons encode circadian feeding time, Nature Neuroscience (2023). DOI: 10.1038/s41593-023-01482-6
© 2023 Science X Network