This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Regional variations uncovered in the impact of dexamethasone treatment for severe COVID-19
There may be important regional differences in the response to higher doses of steroids (dexamethasone) for patients with severe COVID-19, according to a recent sub-study of the COVID-STEROID 2 trial.
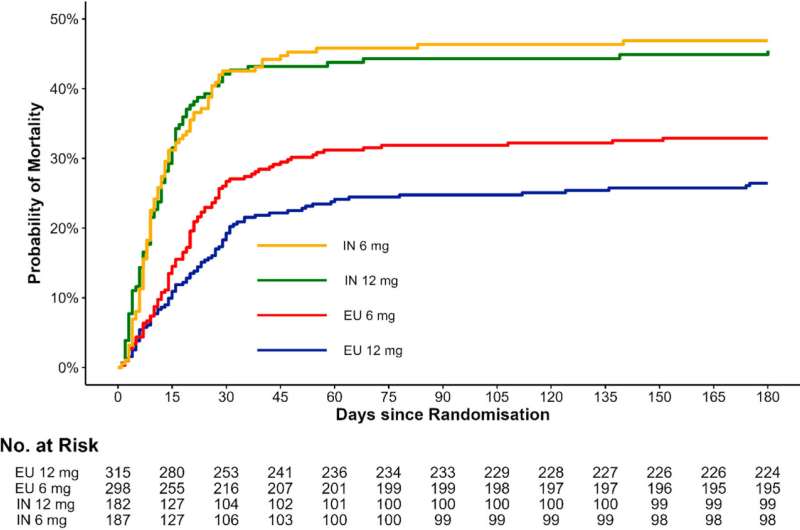
The original COVID-STEROID 2 trial and the current sub-study are a collaboration between The George Institute for Global Health, India and Danish investigators based at CRIC. This study was conducted in Europe and India to examine the effects of giving patients injectable 12 mg versus 6 mg of dexamethasone every day for a maximum of 10 days.
In a secondary analysis of the COVID-STEROID 2 trial published in The Lancet Regional Health—Southeast Asia, the study led by Dr. Bharath Kumar Tirupakuzhi Vijayaraghavan from The George Institute for Global Health India and Apollo Hospital, Chennai highlights the possibility that a patient's geographic location may affect how well this treatment for severe COVID-19 patients works.
Conducting such collaborative research that explores how patients in India and Europe respond differently to higher doses of dexamethasone not only emphasizes the value of international collaboration in large-scale clinical trials related to COVID but also provides a useful way to generate data demonstrating variations in COVID research across various global populations. It is crucial to emphasize the role that these studies play in promoting sustainable global collaborative research projects.
Detailed findings
- Baseline Characteristics: Descriptive baseline data broken down by treatment allocation and geographic region for the 982 patients in the ITT sample. It's interesting to note that baseline variables were largely comparable among treatment arms across areas. There were some significant variations between the two areas, though, such as India's lower weight and its higher use of antivirals and other therapies; also the higher proportion of patients with Diabetes in India.
- Mortality Rates: Patients in Europe and India had differing death rates at various time points, according to the study. On day 28, the higher-dose group's risk of death was 8.3% lower for patients in Europe, whereas there was essentially no difference (0.1%) in India. Comparable patterns were noted on days 90 and 180.
- Serious Adverse Reactions: Regarding serious adverse reactions (SARS), an interesting finding was made. While a larger percentage (-1.0%) of patients in Europe experienced SARS, fewer patients in India suffered SARS at the higher dose (-5.3%).
- Days Alive Without Life Support: The study also revealed variations in the number of days patients survived at day 90 without requiring life support between Europe and India. Patients in India experienced 1.7 more days without life support, compared to 6.1 more days for those in Europe.
- Health-Related Quality of Life (HRQoL): Longer-term HRQoL outcomes were mixed. The adjusted mean difference in EQ5D-5L index values was slightly different between the two regions, with a 0.08 difference in Europe and a 0.02 difference in India. For EQ VAS, the adjusted mean difference was 4.4 in Europe compared to 2.6 in India.
Variations in patient characteristics, health care infrastructure, resource availability, and comorbidity profiles may be the cause of the regional disparities in treatment effects. The observed discrepancies could be explained by variables including different levels of health care-associated infections, a higher incidence of diabetes, and the use of other anti-inflammatory medications such IL-6 inhibitors.
The COVID-STEROID 2 clinical trial examined the effects of two doses of dexamethasone in patients with severe COVID-19 and was carried out in Denmark, India, Sweden, and Switzerland.
More information: Bharath Kumar Tirupakuzhi Vijayaraghavan et al, Heterogeneity of treatment effect of higher dose dexamethasone by geographic region (Europe vs. India) in patients with COVID-19 and severe hypoxemia—a post hoc evaluation of the COVID STEROID 2 trial, The Lancet Regional Health—Southeast Asia (2023). DOI: 10.1016/j.lansea.2023.100293



















