This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Targeting annexin-A1 can halt cancer cell growth, study finds
A new study published in Oncogene highlights the effectiveness of MDX-124, the first therapeutic drug to target annexin-A1, a protein that is overexpressed in several cancer types and promotes tumor progression.
The research was led by Professor Chris Parris and Dr. Hussein Al-Ali at Anglia Ruskin University (ARU) in collaboration with Professor Chris Pepper of Brighton and Sussex Medical School and UK biotech company Medannex, which has produced the MDX-124 monoclonal antibody therapy.
High annexin-A1 expression levels correlate with poorer overall survival in various cancers that currently have limited treatment options, including triple-negative breast, pancreatic, colorectal, and prostate cancers.
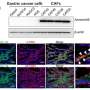
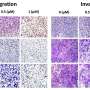
The new study found that MDX-124, which is being developed for use in immunotherapy, significantly reduces proliferation across a number of human cancer cell lines expressing annexin-A1. This anti-proliferative effect is instigated by stopping cell cycle progression.
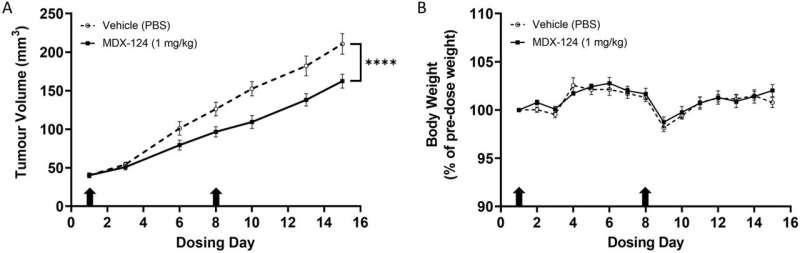
Additionally, MDX-124 is shown to significantly inhibit tumor growth in in vivo models of triple-negative breast and pancreatic cancer, indicating that annexin-A1-targeted therapy represents a viable and innovative approach to cancer treatment.
The phase Ib clinical study of MDX-124, called ATTAINMENT, is currently underway to establish the safety and optimum dose of the novel therapy. Its clinical efficacy will then be evaluated in newly diagnosed cancer patients in combination with current appropriate treatments.
Professor Chris Parris, Head of the School of Life Sciences at Anglia Ruskin University (ARU), said, "We know that the protein annexin-A1 activates formyl peptide receptors to initiate a complex network of intracellular signaling pathways, which can lead to numerous cellular responses, including tumor initiation and progression."
"We have demonstrated in this new study that using MDX-124 can reduce cell growth in annexin-A1-expressing cancer cells both in vitro and in vivo, providing further evidence that annexin-A1 is a valid target for therapy in cancer."
Medannex Director of Scientific Operations, Dr. Fiona Dempsey, who co-authored the paper, said, "We are delighted to publish this work with our collaborators demonstrating the anti-cancer potential of our innovative antibody therapeutic and look forward to the clinical data coming out of the ATTAINMENT study in due course."
More information: Hussein N. Al-Ali et al, A therapeutic antibody targeting annexin-A1 inhibits cancer cell growth in vitro and in vivo, Oncogene (2024). DOI: 10.1038/s41388-023-02919-9