This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Method that rapidly predicts breast cancer survival rates clinically validated
Digistain, a company with its roots at Imperial, has proven the worth of its breast cancer assessment method in a significant clinical trial.
A method that rapidly predicts breast cancer survival rates, based on a technique invented at Imperial, has been successfully tested in a clinical validation study. Patients with a low risk of disease recurrence were identified quickly and accurately, information that would allow nearly half to avoid chemotherapy after surgery.
This novel approach is underpinned by a biopsy imaging technique invented in the Department of Physics at Imperial, which was subsequently developed into a new diagnostic tool by the biotech company Digistain. This tool promises to match the performance of genetic risk profiling methods, without the time and costs involved.
This is borne out in the validation study, published in the journal Breast Cancer Research and Treatment. "The clinical evidence presented demonstrates that this ingenious new approach can deliver accurate results to differentiate high-risk from low-risk breast cancer tumors," said Professor Carlo Palmieri, Consultant in Medical Oncology at the Clatterbridge Cancer Center NHS Foundation Trust, and a co-author of the paper.
"It could be a potential game-changer, given the ease with which the test can be undertaken," he added. This is because the Digistain method is much faster than genetic methods, which require biopsy samples to be processed in specialist labs, often outside the UK.
"Digistain offers hospitals the ability to analyze their own breast cancer biopsy samples for risk recurrence assessment, without the need for special chemical processes," said Professor Palmieri. "This is an exciting and significant technology, with real social impact."
Visibly more efficient
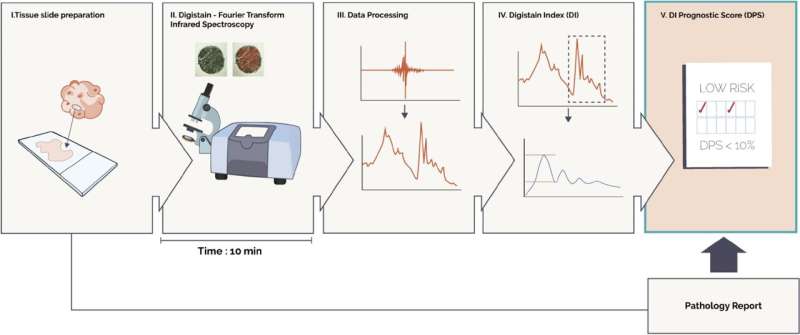
The original imaging technique was developed by Dr. Hemmel Amrania and Professor Chris Phillips in the Department of Physics at Imperial. It analyzes tumor biopsy samples by measuring unique spectroscopic signatures that highlight the chemical changes linked to the onset of cancer. Analyzed via artificial intelligence, these signatures allow the severity of the disease to be established.
This technology was patented and work began to develop a prototype for use by cancer oncologists and pathologists. Dr. Amrania then founded the company Digistain to commercialize the technology and create a tool tailored to breast cancer diagnosis and decision-making.
In the present study, the Digistain test was used to analyze biopsies from more than 800 early-stage breast cancer patients. It was able to classify patients as low or high risk of the cancer returning the next ten years, with an accuracy and predictive performance similar to that reported by other risk stratification tools. Just under half of the patients were flagged as low-risk for disease recurrence.
Existing breast cancer recurrence risk tests are slow, expensive and often involve sending samples overseas, predominantly to the U.S..
"This new technology speeds up a system that is currently too slow and where time and swift action are precious commodities," said Dr. Amrania, Digistain's chief executive.
"This tool will ultimately save lives, save money and prevent patients from the pain and anxiety of needlessly having to wait weeks for results. Importantly, the technology can also reduce the overuse of toxic chemotherapy in many patients."
More information: Charles Coombes et al, Performance of a novel spectroscopy-based tool for adjuvant therapy decision-making in hormone receptor-positive breast cancer: a validation study, Breast Cancer Research and Treatment (2024). DOI: 10.1007/s10549-023-07229-y