This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
How does type 1 diabetes alter muscle structure and blood supply?
In a recent study conducted by the Institute of Anatomy, Faculty of Medicine, University of Ljubljana, researchers have provided new insights into the detrimental effects of type 1 diabetes mellitus (T1DM) on skeletal muscle structure and capillary networks. Utilizing state-of-the-art 3D imaging technology, this comprehensive study marks a significant leap in understanding the multifaceted impact of T1DM on the body's muscular system.
Diabetes mellitus disrupts the regulation of glucose levels, leading to high blood sugar and a myriad of related health issues. T1DM, characterized by the immune-mediated destruction of insulin-producing pancreatic β cells, has profound effects on various organs, especially skeletal muscles, which play a crucial role in glucose uptake and regulation.
This study, published in the journal Biomolecules and Biomedicine, aimed to explore the structural and functional adaptations of skeletal muscles to the metabolic disturbances caused by T1DM.
The hidden changes in muscle and blood vessels
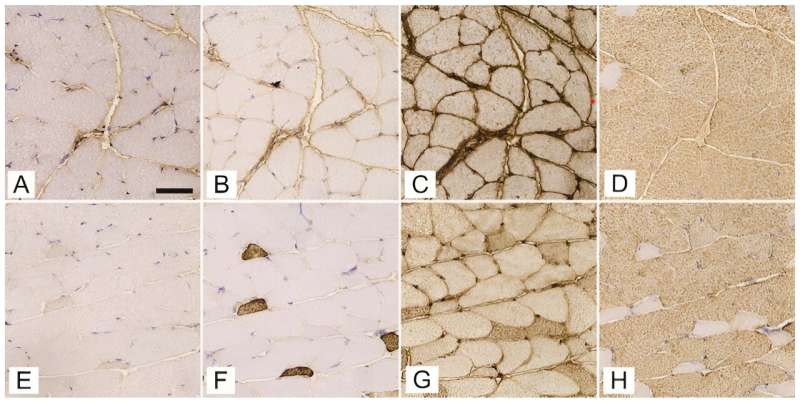
Conducted on female C57BL/6J-OlaHsd mice using a streptozotocin (STZ)-induced model to simulate T1DM, the research focused on critical muscles like the soleus, gluteus maximus, and gastrocnemius. Researchers meticulously analyzed the expression of myosin heavy chain (MyHC) isoforms and the intricacies of the 3D capillary network.
"Our study provides a deeper understanding of how type 1 diabetes not only affects muscle fiber composition but also significantly alters the capillary networks that are essential for muscle health," explained Nejc Umek, the study's lead author.
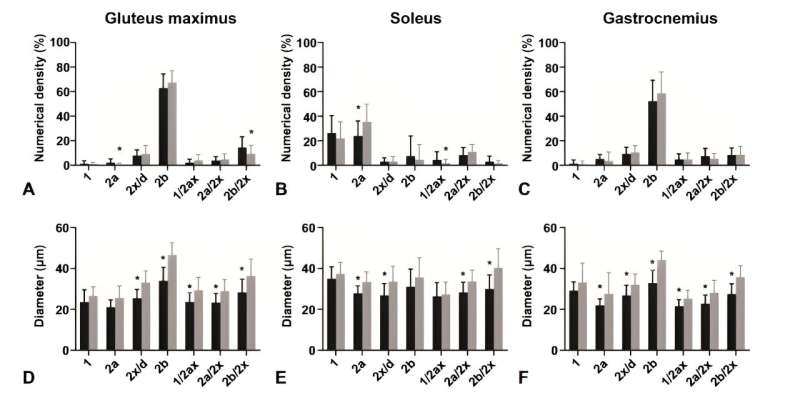
The research revealed that, despite the composition of fast-twitch type 2b fibers remaining consistent, notable differences were observed in the diabetic mice's soleus muscle, which showed a reduced proportion of type 2a fibers and diminished fiber diameters across all muscles analyzed.
Additionally, an intriguing increase in capillary length per muscle volume was discovered in the gluteus maximus of diabetic mice, suggesting an adaptive mechanism to counterbalance muscle fiber atrophy induced by diabetes.
Methodological advances and key discoveries
The study utilized female mice, addressing a gap in diabetes research that often overlooks gender differences in disease progression and response to treatment. Through a single intraperitoneal administration of STZ, researchers successfully induced T1DM, confirmed by significantly elevated fasting glucose levels. This model allowed for an in-depth examination of diabetes-induced changes in a controlled environment.
By employing antibodies specific to different MyHC isoforms and cutting-edge 3D imaging, the team could precisely quantify changes in muscle fiber types and the capillary network. "The advanced 3D imaging techniques we used represent a significant improvement over traditional 2D analyses, offering a more detailed and accurate depiction of the capillary network changes in diabetic muscle tissue," stated Erika Cvetko, the study's senior author.
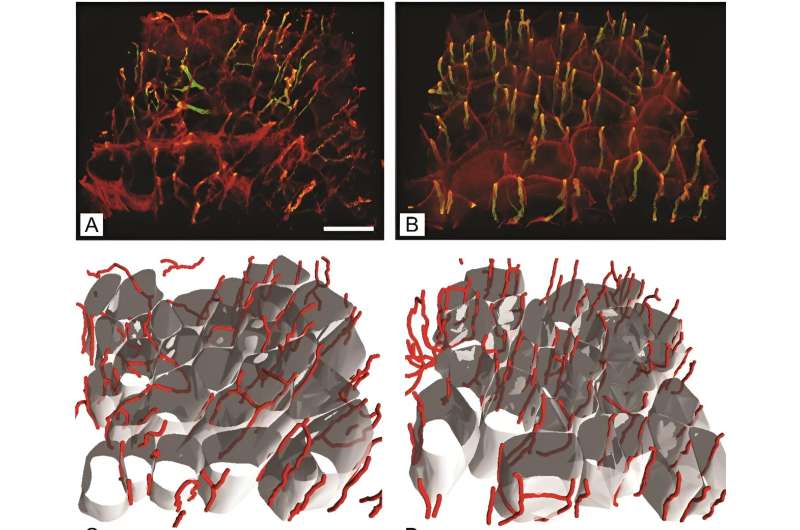
Implications for diabetes management and future directions
The findings from this collaborative research effort highlight the necessity for comprehensive diabetes management plans that encompass not only glucose regulation but also the preservation of muscle structure and function. "Understanding the specific alterations in muscle tissue due to type 1 diabetes paves the way for developing targeted therapies that could significantly improve patient outcomes," Cvetko added.
The study's revelations about the increased capillary length per muscle volume in diabetic mice underscore the body's potential compensatory responses to the structural changes induced by diabetes. These insights are crucial for designing interventions that aim to mitigate muscle deterioration and enhance overall diabetes care.
This novel study contributes significantly to the body of knowledge on diabetes and its systemic effects, particularly on skeletal muscle health. By highlighting the critical role of maintaining muscle integrity and vascular supply in the management of T1DM, the research opens new avenues for therapeutic strategies and underscores the importance of multidisciplinary approaches in tackling this complex disease.
More information: Nejc Umek et al, Skeletal muscle myosin heavy chain expression and 3D capillary network changes in streptozotocin-induced diabetic female mice, Biomolecules and Biomedicine (2023). DOI: 10.17305/bb.2023.9843