This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New epigenetic clocks reinvent how we measure age
What causes us to age? New "clocks" developed by researchers may help point to the answers. Investigators from Brigham and Women's Hospital, a founding member of the Mass General Brigham health care system, unveil a new form of epigenetic clock—a machine learning model designed to predict biological age from DNA structure.
The novel model distinguishes between genetic differences that slow and accelerate aging, predicts biological age and evaluates anti-aging interventions with increased accuracy. Results are published in Nature Aging.
"Previous clocks considered the relationship between methylation patterns and features we know are correlated with aging, but they don't tell us which factors cause one's body to age faster or slower. We have created the first clock to distinguish between cause and effect," said corresponding author Vadim Gladyshev, Ph.D., a principal investigator in the Division of Genetics at BWH. "Our clocks distinguish between changes that accelerate and counteract aging to predict biological age and assess the efficacy of aging interventions."
Aging researchers have long acknowledged the link between DNA methylation—alterations to our genetic structure that shape gene function—and its influence on the aging process. Notably, specific regions of our DNA, known as CpG sites, are more strongly associated with aging. While lifestyle choices, like smoking and diet, influence DNA methylation, so does our genetic inheritance, explaining why individuals with similar lifestyles may age at different rates.
Existing epigenetic clocks predict biological age (the actual age of our cells rather than chronological) using DNA methylation patterns. However, until now, no existing clocks have distinguished between methylation differences that cause biological aging and those simply correlated with the aging process.
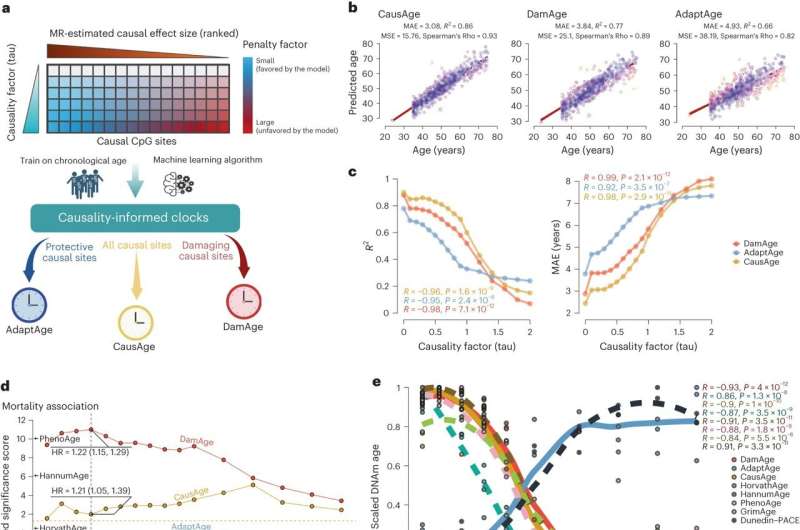
Using a large genetic data set, first author Kejun (Albert) Ying, a graduate student in the Gladyshev lab, performed an epigenome-wide Mendelian Randomization (EWMR), a technique used to randomize data and establish causation between DNA structure and observable traits, on 20,509 CpG sites causal to eight aging-related characteristics.
The eight aging-related traits included lifespan, extreme longevity (defined as survival beyond the 90th percentile), health span (age at first incidence of major age-related disease), frailty index (a measure of one's frailty based on the accumulation of health deficits during their lifespan), self-rated health, and three broad aging-related measurements incorporating family history, socioeconomic status, and other health factors.
With these traits and their associated DNA sites in mind, Ying created three models, termed CausAge, a general clock that predicts biological age based on causal DNA factors, and DamAge and AdaptAge, which include only damaging or protective changes. Investigators then analyzed blood samples from 7,036 individuals ages 18 to 93 years old from the "Generation Scotland Cohort" and ultimately trained their model on data from 2,664 individuals in the cohort.
With these data, researchers developed a map pinpointing human CpG sites that cause biological aging. This map allows researchers to identify biomarkers causative to aging and evaluate how different interventions promote longevity or accelerate aging.
Scientists tested their clocks' validity on data collected from 4,651 individuals in the Framingham Heart Study and the Normative Aging Study. They found that DamAge correlated with adverse outcomes, including mortality, and AdaptAge correlated with longevity, suggesting that age-related damage contributes to the risk of death while protective changes to DNA methylation may contribute to a longer lifespan.
Next, they tested the clocks' ability to assess biological age by reprogramming stem cells (transforming specialized cells, like skin cells, back into a younger, less defined state where they can develop into various types of cells in the body). When applying the clocks to the newly transformed cells, DamAge decreased, indicating a reduction in age-related damage during reprogramming, while AdaptAge did not show a particular pattern.
Finally, the team tested their clocks' performance in biological samples from patients with various chronic conditions, including cancer and hypertension, as well as samples damaged from lifestyle choices like smoking cigarettes. DamAge consistently increased in conditions associated with age-related damage, while AdaptAge decreased, effectively capturing protective adaptations.
"Aging is a complex process, and we still do not know what interventions against it actually work," said Gladyshev. "Our findings present a step forward for aging research, allowing us to more accurately quantify biological age and evaluate the ability of novel aging interventions to increase longevity."
More information: Kejun Ying et al, Causality-enriched epigenetic age uncouples damage and adaptation, Nature Aging (2024). DOI: 10.1038/s43587-023-00557-0