This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Mapping potential pathways to motor neuron disease treatment
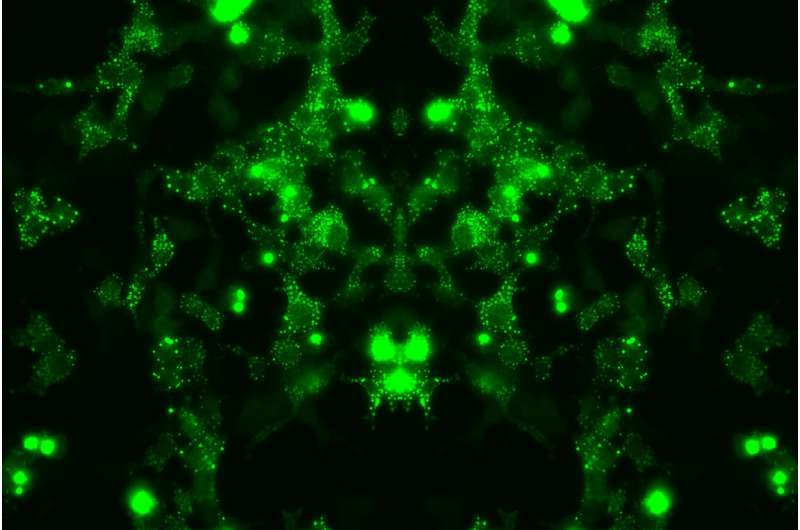
For the first time, researchers from the University of Queensland (UQ) have mapped out the proteins implicated in the early stages of motor neuron disease (MND).
Dr. Rebecca San Gil from Associate Professor Adam Walker's lab at UQ's Queensland Brain Institute has developed a longitudinal map of the proteins involved in MND across the trajectory of the disease, identifying potential therapeutic pathways for further investigation.
"The map is a springboard for many more projects exploring the proteins activated and repressed during the onset, early and late stages of MND," Dr. San Gil said.
"These proteins are biological factors that drive disease onset and progress its development over time. We measured differences in protein levels in the brain across the trajectory of the disease and collated this information into a longitudinal map."
The paper is published in Nature Communications. The map is now available for scientists worldwide and will accelerate investigations into MND.
Dr. San Gil has been working in mouse models of MND to understand the mechanisms driving TDP-43 pathology in the brain, which accounts for 95% of amyotrophic lateral sclerosis (ALS) cases and 50% of frontotemporal lobar degeneration (FTLD).
Building on the mapping project, Dr. San Gil chose to focus on a protein-folding factor called DNAJB5.
"Before the onset of MND in mouse models, we observed a marked increase in protein groups responsible for physically assisting in the protein folding process.
"One of these 'chaperone' proteins, DNAJB5, was particularly abundant early on, sparking our curiosity about its role in disease progression.
"In human brain tissue, we found DNAJB5 enriched in areas where TDP-43 aggregates.
"The short-term elevation of DNAJB5 is likely a protective mechanism by neurons in an attempt to control TDP-43 as it begins to dysfunction.
"This protective response to TDP-43 needs further investigation because it may help us identify preventative and therapeutic approaches to MND."
A/Prof. Walker envisions that the lab will continue to follow other identified protein pathways, using gene therapy and repurposing medicine, to see if they can alter or prevent the disease.
More information: Rebecca San Gil et al, A transient protein folding response targets aggregation in the early phase of TDP-43-mediated neurodegeneration, Nature Communications (2024). DOI: 10.1038/s41467-024-45646-9