This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New screening tool to explore mechanisms behind cancer, autoimmunity and neurodegeneration
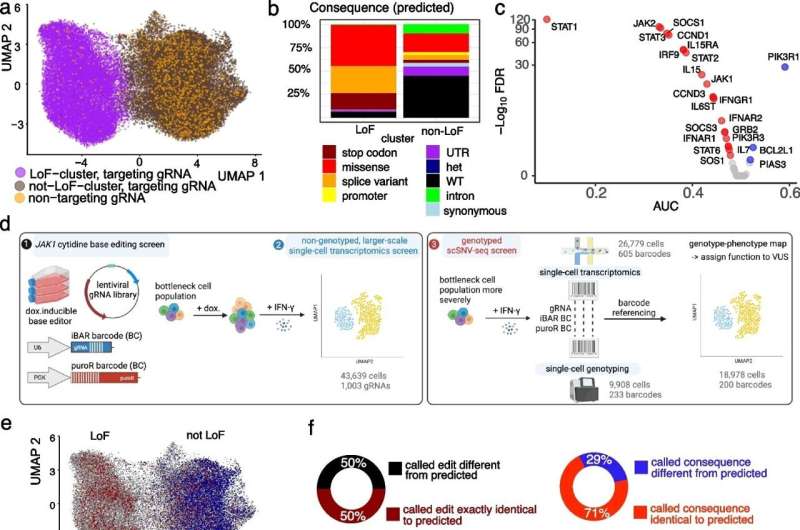
Scientists have developed a new screening tool to uncover how genetic changes affect gene activity and can lead to diseases such as cancer, autoimmunity, neurodegeneration and cardiovascular disease. This new tool enables the investigation of thousands of DNA mutations identified by genetic studies in one experiment, guiding the development of advanced diagnostics and treatments.
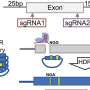
The technique, called scSNV-seq, enables researchers to rapidly assess the impact of thousands of genetic changes in cells that have never been screened before, directly connecting these changes to how those same cells operate. This provides a comprehensive view from which researchers can pinpoint the mutations that contribute to disease. This will offer crucial insights for developing targeted therapies.
In the new study published in Genome Biology, researchers from the Wellcome Sanger Institute and their collaborators at Open Targets and EMBL's European Bioinformatics Institute (EMBL-EBI) applied scSNV-seq to the blood cancer gene JAK1.
The technique accurately assessed the impact of JAK1 mutations, revealing for the first time that certain mutations caused a "halfway house" phenotype cycling between different states. This is not possible under previous approaches.
The technique is designed to demonstrate versatility across cell types, including hard-to-culture primary cells like T cells and stem-cell-derived neurons, as well as various editing methods such as base editing and prime editing. Applied on a large scale, scSNV-seq could transform understanding of the genetic changes driving cancer and decoding genetic risk for Alzheimer's, arthritis, diabetes, and other complex diseases.
Advances in human genetics combined with the increasing affordability of DNA sequencing technologies have unveiled hundreds of thousands of disease-related genetic variants that are increasing at a staggering rate. Yet, tools to interpret them lag behind, sometimes relying on tedious manual processes.
When using advanced gene-editing tools to introduce defined genetic mutations, using current screening methods, it is difficult to distinguish between cells where the editing did not work and those where it successfully introduced a harmless change without affecting the cell's behavior.
Researchers from the Wellcome Sanger Institute and their collaborators set out to address this with a new screening technique, scSNV-seq, which directly couples the specific genetic information in the genotype of a cell to its gene activity. The team tested the effectiveness of scSNV-seq by altering specific DNA bases within the JAK1 gene, which is linked to inflammation and cancer, to study their effects on cell behavior.
They demonstrated scSNV-seq could accurately categorize different types of genetic changes into three categories: benign, causing loss of function, and altering function. They showed certain mutations caused an intermediate phenotype cycling between different states—an observation not possible under existing approaches.
Dr. Sarah Cooper, first author of the study at the Wellcome Sanger Institute, said, "In an era where the rate of genetic variant discovery outpaces our ability to interpret their effects, scSNV-seq fills a major gap for studying challenging cells like T cells and neurons. We are already using it to shed light on the impact of Alzheimer's and Parkinson's risk variants on brain cells."
Dr. Andrew Bassett, senior author of the study at the Wellcome Sanger Institute, said, "Our technique is able to directly connect effects of mutations to how a cell behaves, revealing downstream impacts that previous technologies alone cannot deliver. The technique speeds up the identification of causal genetic mutations, which will allow better diagnosis and deepens our molecular understanding of diseases, paving the way for more targeted and effective treatments."
More information: Sarah E. Cooper et al, scSNV-seq: high-throughput phenotyping of single nucleotide variants by coupled single-cell genotyping and transcriptomics, Genome Biology (2024). DOI: 10.1186/s13059-024-03169-y