This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Two new mutations identified as possible causes of rare blood disorder
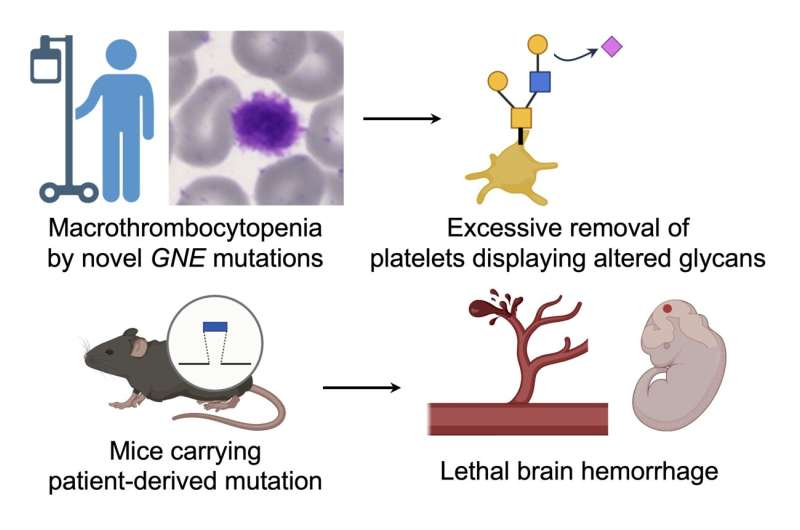
Researchers have uncovered two novel gene mutations that may be responsible for a rare blood disorder. The mutations are found on the GNE gene and impact an enzyme, also called GNE, that is involved in the synthesis of sialic acid and a process called sialylation, which is critical for brain development. The mutations were first identified in a pediatric patient with macrothrombocytopenia, also called a giant platelet disorder, because of the enlarged size of blood platelets.
The findings were reported in a paper published in Blood Advances.
"We identified two novel mutations (C594Y and P735R) in the GNE gene, which is required for sialic acid biosynthesis. By making mice carrying P735R of patient-derived mutations, we set out to determine if GNE mutations cause macrothrombocytopenia. This is the first study confirming that GNE mutations cause platelet problems in mice," said co-first author Yuji Kondo, Ph.D., Lecturer at the Institute for Glyco-core Research (iGCORE) at Nagoya University in Nagoya, Japan.
Once the mutations were identified, researchers replicated one of the mutations (P735R) in a mouse model because they predicted that it was more likely to alter GNE.
The mice that were given this mutation died as embryos after fatal brain bleeds. In previous research, mice that have been given other GNE mutations survived after birth, so this was not an expected result. Though unexpected, the death of the mouse embryos with the P735R GNE mutation has provided researchers with some answers and more questions for future research.
The death of the mice embryos with the P735R mutation reveals the role of the GNE enzyme and sialylation in angiogenesis during embryonic brain development. Angiogenesis is the process that forms new blood vessels.
"Our body has two pathways for sialic acid synthesis. One is a GNE-mediated 'de novo' sialic acid biosynthetic pathway. The other is a 'salvage' pathway from lysosomal degradation of glycoconjugates."
"Our study indicates that the GNE-mediated de novo sialic acid biosynthetic pathway is critical for embryonic development, and the salvage pathway and supply of sialic acid from the pregnant mother through the placenta to the fetus are not sufficient for development," said Kondo. In simple terms, the P735R mutation stops sialylation via the GNE-mediated pathway, and the salvage pathway cannot make up the slack, causing defective angiogenesis in the developing embryo.
The researchers predict that mice with the other newly discovered mutation, C594Y, will live longer and provide additional opportunities to study these genetic mutations and how they impact blood disorders.
"Our next step is to analyze phenotypes of C594Y-carrying mice to see if they have the platelet problem. If so, we will analyze the mechanism underlying macrothrombocytopenia and find approaches for therapeutic intervention. We will also seek to learn more about the myopathy phenotype and identify the molecular mechanism for how GNE myopathy develops and what factors contribute to fine-tuned myopathy development," said Kondo.
More information: Lulu Huang et al, Novel GNE missense variants impair de novo sialylation and cause defective angiogenesis in the developing brain in mice, Blood Advances (2024). DOI: 10.1182/bloodadvances.2023011490