This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Eating, socializing or exploring: How the brain switches between different behaviors
How does our brain switch between different behaviors? A new study has now provided the first answers to this key question in neuroscience. Using mice, the researchers investigated electrical activity in a certain area within the brain. Results were then analyzed with the help of an adaptive computer algorithm. This artificial intelligence identified a type of typical fingerprint in the signals.
Analyzing this signal allowed researchers to predict which behavior the animals would switch to next, two seconds before they actually made the change. The results have now been published in the journal Nature Neuroscience.
The study was conducted at Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), the University Clinic of Cologne and the Max Planck Institute for Metabolism Research.
We do thousands of different things every day: we phone, we write emails, we eat, we play sports, we brush our teeth. How does our brain manage to switch between these different behaviors? This question is currently the subject of intense research in neuroscience. If this central function of our brain fails to work properly, it can lead to major neuropsychiatric disorders, such as binge eating, anorexia or obsessive–compulsive disorder.
A certain area within the brain plays an important role in making these transitions: the hypothalamus. Its role is akin to that of an air traffic controller. It acts as a hub where important information about the body comes together—whether we are hungry, how high our body temperature is, how fast our heart is beating.
The rest of the brain keeps the hypothalamus informed about the external world. Based on this information, the hypothalamus regulates innate behaviors such as eating, exploring surroundings or interacting with others. But how does it do so?
To answer this question, a tandem of research groups lead by Prof. Dr. Alexey Ponomarenko at the Institute of Physiology and Pathophysiology at FAU and Prof. Dr. Tatiana Korotkova at the University Clinic of Cologne and the MPI for Metabolism Research combined several cutting-edge techniques of neuroscience and mathematics.
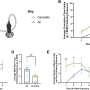
The researchers investigated the hypothalamus in mice, as this part of the brain is very similar in mice and humans. "We used AI to analyze the electrical activity in a certain region within the hypothalamus," explains data scientist Mahsa Altafi, a doctoral candidate at FAU.
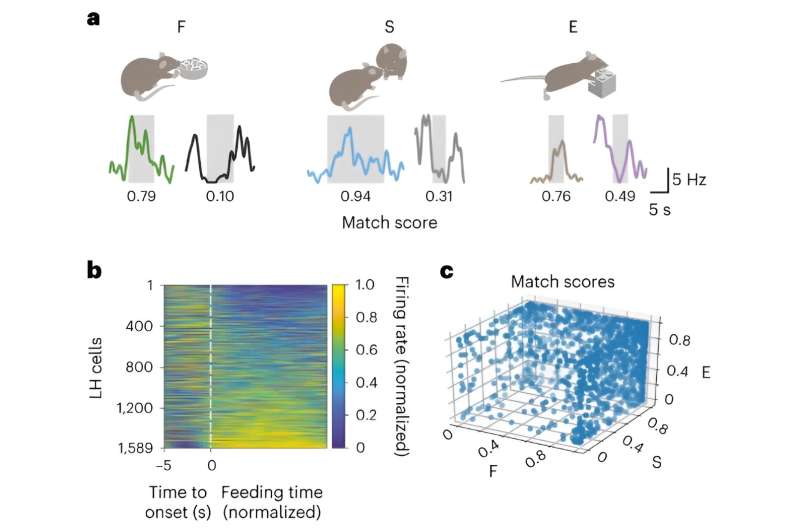
Initial findings revealed that the hypothalamus oscillates at a rhythm known as a beta oscillation. The nerve cells in the hypothalamus are particularly active 20 times a second, with activity levels falling again between these peaks. It is like an orchestra in which all the musicians concentrate on the conductor's baton in order to play in unison.
What is particularly interesting is that some cells do not become active on tact, but just before, in the offbeat. These cells play a certain melody. And the order of the notes they play influences which piece of music the orchestra plays next. "Reading the electrical signal allows us to predict which behavior the mouse will switch to two seconds later," explains Altafi.
But what happens if the offbeat melody is suppressed? Changwan Chen at the Max Planck Institute for Metabolism Research and the University Clinic of Cologne used light to manipulate the activity of the hypothalamic neurons.
The effect was surprising: The mice remained stuck in their current behavior until the light was switched off. For example, they interacted persistently with other mice even if these mice showed no interest. "It was striking how persistently a mouse with the inhibited transition state interacted with another mouse that was trying to avoid this prolonged communication," Chen recalls.
The "offbeat melody" appears to put the hypothalamus in a transition state, thereby enabling the animals to switch to another behavior. Which behavior that is, however, does not depend solely on the hypothalamus.
It turns out that the hypothalamus is instructed by the medial prefrontal cortex, a region responsible for cognitive control of behavior. For example, it considers, which option is best in a particular situation. Should I eat? Or should I rather interact with another mouse, or collect new experiences?
In order to communicate with the hypothalamus, the medial prefrontal cortex oscillates in tact with the rhythm set by the hypothalamus, following the beta oscillation like a conductor's baton. "Signals from the prefrontal cortex help the hypothalamus to promote transitions between behaviors," explains Prof. Korotkova from the University Clinic of Cologne.
"It is particularly fascinating that the hypothalamus starts a preparation to transition between behaviors around two seconds before it actually occurs. It is likely that the mice are not even consciously aware at this point that they are about to switch to a different behavior."
"Our findings indicate the importance of beta oscillations in orchestrating the activity of the myriad of neurons that drive specific behaviors, and for smooth transitions between them," states Prof. Ponomarenko.
"These findings may guide the development of new medications and therapies for serious psychiatric brain disorders. I look forward to the day when patients with anorexia nervosa or obsessive-compulsive disorder can benefit from this."
More information: Changwan Chen et al, The dynamic state of a prefrontal–hypothalamic–midbrain circuit commands behavioral transitions, Nature Neuroscience (2024). DOI: 10.1038/s41593-024-01598-3. www.nature.com/articles/s41593-024-01598-3