This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study finds specific brain wiring in mice that controls getting ready for bed

A team, led by Imperial College London researchers, has uncovered the wiring in mouse brains that leads them to begin nesting in preparation for sleep. Published in Nature Neuroscience, the study reveals that preparing properly for sleep is likely a hard-wired survival feature—one often neglected or overridden by humans.
We all need to sleep, but since we are unconscious when we do so, it makes sense to fall asleep in a safe and warm place. For some animals this is especially important, as a burrow or nest provides a haven from predators.
Dr. Kyoko Tossell, from the Department of Life Sciences at Imperial, and colleagues in the Franks-Wisden lab found that depriving mice of sleep produces an instinctive behavior to build a nest, or in other words, get ready for bed. They also revealed the brain wiring responsible for this behavior, which is likely to be shared across mammals.
Dr. Tossell said, "Although it seems to us humans that we have a conscious control of the process of going to bed, it is very likely that the same wiring exists in the human brain as the mouse brain. So, even if we think we are making a choice, we are probably governed by unconscious urges to prepare for bed as mice are."
Professor Bill Wisden, Chair in Molecular Neuroscience at Imperial, added, "Getting a good night's sleep is vital for health. And preparing for bed properly—what clinicians refer to as 'sleep hygiene' (such going to bed at a suitable time, on a proper mattress, with good sheets and no phone or TV)—is equally important, but is often neglected or overridden by many."
"The fact that this process is hard-wired in mice shows that preparing properly for sleep is a survival feature used in nature, and so humans should take this aspect of their sleep seriously."
To delve deeper into the sleep preparations of mice, the team deprived the mice of sleep and then observed their behavior and brain activity when they were eventually allowed to drop off. They made mice sleepy by presenting them with Lego bricks and other toys every half an hour for five hours, which caused them to stay awake instead of napping, and filmed their nesting behavior before they finally went to sleep.
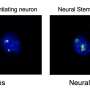
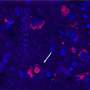
To look at the brain wiring involved in nesting, the authors used a method called optogenetics, where particular brain cells can be artificially activated by a flash of light. This allowed them to experimentally verify that dedicated brain cells were responsible for nesting when the mice were sleepy.
They discovered that sleep preparatory behavior is wired up in the top of the brain, in a region called the prefrontal cortex. In humans and mice, the prefrontal cortex helps in rational planning, or 'executive control," which is often essential for survival.
As mice become progressively more tired, the team discovered that special types of neurons become active in the prefrontal cortex and send signals down to the base of the brain, the hypothalamus. The hypothalamus is the brain's control center for regulating many of the basic survival processes needed by the body, including eating, drinking, and sleeping.
Down in the hypothalamus, special memories (or instincts) are stored for nest building and the wiring to trigger sleep.
Dr. Tossell said, "In other words, when tiredness starts to wash over the mouse, the top part of the brain (the prefrontal cortex) decides to implement behavior to make sure the mouse is in a safe place before going to sleep."
Now this brain wiring has been discovered, the team want to expand their studies to uncover what makes the prefrontal cortex cells active during tiredness and how the brain actually senses tiredness.
Professor Nick Franks said, "As the next steps for our work, we would like to discover the answers to these questions as they may provide an answer to one of the biggest questions in neuroscience: why do we sleep?"
More information: Somatostatin neurons in prefrontal cortex initiate sleep-preparatory behavior and sleep via the preoptic and lateral hypothalamus, Nature Neuroscience (2023). DOI: 10.1038/s41593-023-01430-4. www.nature.com/articles/s41593-023-01430-4