This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Experts call for more clinical trials on alcohol use, liver disease
More clinical research is needed to investigate how reducing alcohol consumption in patients with alcohol-related liver disease (ALD) may slow disease progression and improve outcomes, according to an international task force of experts from more than two dozen institutions including UT Southwestern Medical Center.
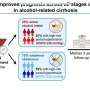
In a consensus statement published in Nature Reviews Gastroenterology & Hepatology, the group, commissioned by the National Institute on Alcohol Abuse and Alcoholism (NIAAA), issued a set of 28 recommendations to guide the design of future clinical trials addressing alcohol use disorder (AUD) and ALD. The recommendations call for multidisciplinary teams, including experts from hepatology and addiction medicine, and the need to address the stigma of alcoholism.
"Physicians know anecdotally that reducing or eliminating heavy drinking after diagnosis can extend and improve life for patients with ALD, but research on the subject has been limited," said Mack Mitchell, M.D., Professor of Internal Medicine in the Division of Digestive and Liver Diseases and Vice President for Medical Affairs at UT Southwestern, who co-chaired the task force and is the lead author on the article. "Our hope is that well-designed clinical trials focused on alcohol use and ALD could lead to effective changes in clinical practice."
Deaths linked to excessive alcohol use have increased over the past two decades, including a surge during the COVID-19 pandemic. Alcohol consumption is a primary cause of death from liver-related diseases, accounting for more than half of all cirrhosis-related deaths in the U.S. in 2022 and 46% of all liver disease deaths, according to NIAAA. While abstinence has been associated with improved life expectancy for ALD patients, few clinical trials have been conducted to investigate how interventions to reduce or eliminate heavy drinking will affect disease progression.
The task force included hepatologists, addiction medicine specialists, clinical trialists, and members of regulatory agencies. Among its recommendations are definitions for disease categories, criteria for trial participants, treatment considerations, and safety and regulatory concerns.
The article notes that diseases related to alcohol use are among the most stigmatized, resulting in stereotypes that create barriers to treatment, such as ineligibility for liver transplantation. The group proposed that researchers discuss ways to reduce those negative beliefs among all team members in trials for AUD and ALD and, when possible, involve patient representatives and other stakeholders in trial design.
Thomas Cotter, M.D., M.S.C.P., Assistant Professor of Internal Medicine in the Division of Digestive and Liver Diseases at UT Southwestern, also participated in the consensus statement.
"The field of alcohol-associated liver disease lacks well-designed clinical trials integrating alcohol use disorder treatment, a pivotal component in order to optimize patient outcomes. These recommendations will serve as an important blueprint to help guide the field going forward," Dr. Cotter said.
Nature Reviews Gastroenterology & Hepatology has also published a companion commentary on the subject.
More information: Consensus statement: Brian P. Lee et al, Designing clinical trials to address alcohol use and alcohol-associated liver disease: an expert panel Consensus Statement, Nature Reviews Gastroenterology & Hepatology (2024). DOI: 10.1038/s41575-024-00936-x
Commentary: Juliana Serrazina et al, Shaping new paths in clinical trial design to address alcohol use disorders and alcohol-associated liver disease, Nature Reviews Gastroenterology & Hepatology (2024). DOI: 10.1038/s41575-024-00948-7