This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Starving cancer cells to enhance DNA damage and immunotherapy response
A new research perspective titled "Starving cancer cells to enhance DNA damage and immunotherapy response" has been published in Oncotarget.
Prostate cancer (PCa) poses significant challenges in treatment, particularly when it progresses to a metastatic, castrate-resistant state. Conventional therapies, including chemotherapy, radiotherapy, and hormonal treatments often fail due to toxicities, off-target effects, and acquired resistance. In this new research perspective, researchers Aashirwad Shahi and Dawit Kidane from Howard University define an alternative therapeutic strategy focusing on the metabolic vulnerabilities of PCa cells, specifically their reliance on non-essential amino acids such as cysteine.
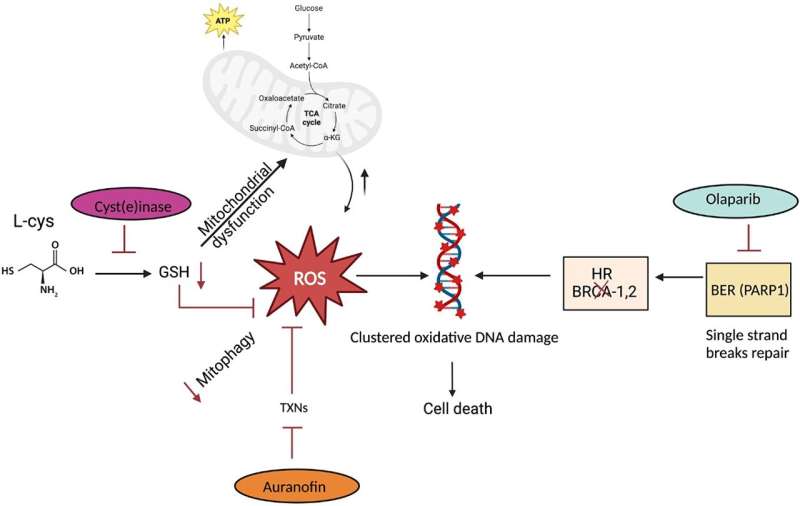
Using an engineered enzyme cyst(e)inase to deplete the cysteine/cystine can induce oxidative stress and DNA damage in cancer cells. This depletion elevates reactive oxygen species (ROS) levels, disrupts glutathione synthesis, and enhances DNA damage, leading to cancer cell death. The combinatorial use of cyst(e)inase with agents targeting antioxidant defenses, such as thioredoxins, further amplifies ROS accumulation and cytotoxicity in PCa cells.
"Overall, this perspective provides a compressive overview of the previous work on manipulating amino acid metabolism and redox balance [to] modulate the efficacy of DNA repair-targeted and immune checkpoint blockade therapies in prostate cancer," state the researchers.
More information: Aashirwad Shahi et al, Starving cancer cells to enhances DNA damage and immunotherapy response, Oncotarget (2024). DOI: 10.18632/oncotarget.28595