Analysis of mTOR shows how the protein works, how new generation of drugs may defeat it
Uncovering the network of genes regulated by a crucial molecule involved in cancer called mTOR, which controls protein production inside cells, researchers at the University of California, San Francisco (UCSF) have discovered how a protein "master regulator" goes awry, leading to metastasis, the fatal step of cancer.
Their work also pinpoints why past drugs that target mTOR have failed in clinical trials, and suggests that a new class of drugs now in trials may be more effective for the lethal form of prostate cancer for which presently there is no cure.
Described this week in the journal Nature, the protein mTOR is a "master regulator" of human protein synthesis. It helps normal cells sense nutrients and control cell growth and metabolism. But in many forms of cancer, this process goes awry, and mTOR reprograms normal cells to aberrantly divide, invade and metastasize.
"Many human cancers show hyperactivation of this pathway," said Davide Ruggero, PhD, an associate professor of urology and member of the Helen Diller Family Comprehensive Cancer Center and the Multiple Myeloma Translational Initiative at UCSF. "Until now, we have not known how hyperactive mTOR perturbs the synthesis of certain proteins leading to fatal cancer."
In the human body, mTOR is a molecular sensor that helps cells respond to favorable or unfavorable environments. Under ordinary conditions, it acts as a master regulator of genes that induce cells to growth and divide. In times of scarcity, when somebody is starving for instance, mTOR shuts down much of the machinery that makes proteins so that an organism can conserve energy.
In cancer, this careful balance is lost. The rogue mTOR protein goes haywire and signals tumor cells to become bigger to divide, undergo metastasis and invade new, healthy tissues. Metastasis is the primary cause of cancer patient death.
"We are now discovering that during tumor formation mTOR leads to metastasis by altering the synthesis of a specific group of proteins that make the cancer cells move and invade normal organs," Ruggero said.
In their research, Ruggero and his colleagues identified the players that instruct or execute decisions made by mTOR, and they discovered how mTOR deregulates one of the very last stages of gene expression—just before they are translated into proteins by large molecular machines known as ribosomes.
They used a method called ribosome profiling pioneered by UCSF professor Jonathan S. Weissman and Nicholas T. Ingolia at the Carnegie Institution for Science, also authors on the paper. This method basically allows researchers to collect the millions of ribosomes from inside cells and determine which genes they are turning into proteins.
The Drug and its Target
Because it plays such a crucial role in cancer biology, mTOR is also an active target for drug development. Several compounds that block this protein, including the drug Rapamycin, have already gone through clinical trials as single agents for treating various forms of cancer such as prostate cancer—without great success.
The new research suggests why drugs like Rapamycin have failed. The problem, said Ruggero, is that they block mTOR, but not completely. The newer drugs, however, block mTOR more completely.
The difference is akin to holding a door shut with a piece of tape versus locking it and breaking the key off in the lock so no one can re-open it.
When drugs like Rapamycin fail to completely stop mTOR from working, they allow it to continue pushing a cancer cell toward malignancy. Some newer compounds that block mTOR do so more completely, and the team led by Ruggero showed in preclinical experiments that this effectively hobbles the cancer cells.
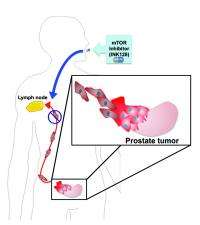
Specifically they tested an experimental drug called INK128, derived from a compound discovered in the UCSF laboratory of Kevan Shokat, Howard Hughes Medical Investigator, professor and chair of cellular and molecular pharmacology and another author on the paper. This compound is now in clinical trials for different types of cancers and is being developed by a team led by Christian Rommel in the La Jolla, Calif. company, Intellikine, Inc., the home of some of the co-authors in the Nature manuscript.
In their research, Ruggero and his colleagues showed that a mouse model of human prostate cancer treated with INK128 did not metastasize. They also showed that the new drug has a strong therapeutic effect on human prostate cancer cells.
"While the experiments were primarily focused in prostate cancer, we believe this work is widely applicable in many tumor types because mTOR is a critical regulator of so many cancers," said first author Andrew Hsieh, MD, a clinical oncologist in the UCSF Department of Medicine, Division of Hematology/Oncology and a senior member of the Ruggero laboratory. "For example, clinicians Jeff Wolf and Tom Martin are now testing INK128 here at UCSF, in multiple myeloma patients," Ruggero said.
The research also found that INK128 works better by also restraining abnormal protein synthesis when mTOR is hyperactive. "Deregulations in protein synthesis is now becoming a hallmark of cancer, and we are very excited by the opportunity to target the aberrant protein synthesis apparatus in many cancers," Ruggero said.
More information: Nature, Advance Online Publication. February 22, 2012 - dx.doi.org/10.1038/nature10912














