Research points to new way of preserving fertility for boys undergoing cancer treatment
(Medical Xpress) -- Treatments for childhood cancers are increasingly successful with cure rates approaching 80%, but success often comes with a downside for the surviving men: the cancer treatments they received as boys can leave them sterile as adults. Now, a research team led by Ralph Brinster of the University of Pennsylvania School of Veterinary Medicine has completed a 14-year experiment that gives hope for a technique that could restore their fertility.
Brinster is the Richard King Mellon Professor of Reproductive Physiology at Penn Vet and was recently awarded the National Medal of Science for his lifetime of research on the genetics of the mammalian germline, the cells that give rise to sperm and eggs.
In his most recent research, Brinster collaborated with fellow members of the Department of Animal Biology at Penn Vet, with members of the Department of Cell and Developmental Biology at Penn’s Perelman School of Medicine and with the Penn Bioinformatics Core.
Their study was published in the journal Human Reproduction.
For males, fertility begins with spermatogonial stem cells, which are present at birth, embedded in the basement membrane of the testes’ seminiferous tubules. As a boy approaches puberty, these cells begin to make daughter cells that eventually become sperm. While they normally continue this process throughout a post-pubescent man’s life, factors like radiation and chemotherapy drugs can destroy them, rendering him sterile.
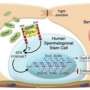
About 1 in 3 boys surviving childhood cancer will be in danger of having severely decreased fertility as an adult; as many as 1 in 5,000 men of reproductive age currently suffer this serious quality-of-life problem as a result. Adult men who undergo cancer treatment that might damage their fertility can preemptively freeze their sperm, an option not available to pre-pubescent boys. But if a sample of a boy’s spermatogonial stem cells could be extracted and preserved before cancer treatment and re-implanted after the boy reached adulthood, this fertility problem could be circumvented.
“There are a number of places, including at the Children’s Hospital of Philadelphia,” Brinster said, “that are already freezing cells for patients to use later, with the expectation that the necessary culture system and implantation techniques will be developed. A logical question for patients to ask is, How do we know that, after 10 years or more of being stored, these cells are any good? That’s what our study addresses.”
The techniques for extracting these cells and re-implanting them have been developed, so a critical question for researchers was whether spermatogonial stem cells could survive the decade-plus period they might need to remain frozen.
Fortunately, Brinster had a large collection of cryopreserved spermatogonial stem cells stored in the mid-’90s. The collection consisted mostly of cells taken from mice but included a small number of rat, rabbit and baboon samples. Between their age and variety, these samples represented an important resource to address questions regarding long-term cryopreservation of spermatogonial stem cells, before the technique could be used in humans.
After being thawed, the spermatogonial stem cells of rabbits and baboons were labeled with a fluorescent dye so that the researchers could track where in the testes they would eventually migrate and embed once they were implanted in mice. This was a critical step for the rabbit and baboon cells, which lacked appropriate experimental recipients; once implanted in mice, the studies showed that the cells migrated correctly and embedded in the recipient testes’ basement membrane, indicating their viability.
A complete functional test could only be done for the mouse spermatogonial stem cells, as they could be implanted into mice and tested to see if they could produce sperm, and whether that sperm could lead to healthy offspring. After demonstrating that the cryopreserved cells implanted in the right region of the testes and underwent normal spermatogenesis, the researchers extracted the sperm and successfully fertilized eggs in vitro.
One of the recipient mice was also placed with females and successfully sired offspring. The offspring resulting from eggs fertilized in vitro as well as those resulting from natural mating all produced normal appearing young. Most importantly, the offspring arising from the cryopreserved spermatogonial stem cells appeared free of genetic damage.
This experiment clearly points in the right direction for the feasibility of a similar fertility treatment in humans.
“Human and animal spermatagonial stem cells have been successfully frozen for short periods of time, but this is completely different,” Brinster said. “Here we had cells frozen for over a decade that implanted in the right place and made sperm, and that sperm made offspring without apparent genetic defects.”