Molecular probes identify changes in fibronectin that may lead to disease
Fibronectin plays a major role in wound healing and embryonic development. The protein, which is located in the extracellular matrix of cells, has also been linked to pathological conditions including cancer and fibrosis.
During physiological processes, fibronectin fibers are believed to experience mechanical forces that strain the fibers and cause dramatic structural modifications that change their biological activity. While understanding the role of fibronectin strain events in development and disease progression is becoming increasingly important, detecting and interrogating these events is difficult.
In a new study, researchers identified molecular probes capable of selectively attaching to fibronectin fibers under different strain states, enabling the detection and examination of fibronectin strain events in both culture and living tissues.
"The mechano-sensitive molecular probes we identified allow us to dynamically examine the relevance of mechanical strain events within the natural cellular microenvironment and correlate these events with specific alterations in fibronectin associated with the progression of disease," said Thomas Barker, an assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
The study was published on April 23, 2012 in the online early edition of the journal Proceedings of the National Academy of Sciences. Barker worked on the study with Georgia Tech graduate student Lizhi Cao and Harry Bermudez, an assistant professor in the University of Massachusetts Amherst Department of Polymer Science and Engineering. The research was supported by the National Institutes of Health.
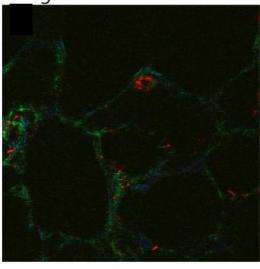
Researchers have hypothesized that mechanical forces emanating from cells may partially unfold fibronectin and regulate what proteins bind to it. While simulation and tissue culture experiments support this hypothesis, direct evidence that such molecular events occur in living organisms has not yet been presented, according to Barker.
A technique called intramolecular fluorescence resonance energy transfer (FRET) has been used to detect molecular strain events in fibronectin fibers, but the technique has limitations because it cannot be used on living tissues and requires the fibronectin to be chemically labeled.
"The molecular probes we identified can be used to map molecular strain events in native extracellular matrix and living lung tissues," explained Barker. "The probes can also be used to study the mechanism by which cells control the mechanical forces that alter fibronectin's conformation, control the exposure of its binding sites and regulate cell signaling."
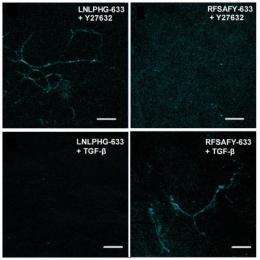
The researchers used a controlled fibronectin fiber deposition and extension technique to apply tension to the fibers and stretch them to 2.6 times their original length without significant breakage. Then they used a technique called phage display to identify peptides capable of discriminating fibronectin fibers under relaxed and strained conditions. The molecular probes displaying peptide sequences LNLPHG and RFSAFY showed the greatest binding affinity to fibronectin fibers and the greatest efficiency in discriminating between relaxed and strained fibers.
For proof-of-concept demonstrations, the researchers used the probes to discriminate fibronectin fibers within native extracellular matrix and mouse lung slices. LNLPHG preferentially attached to relaxed fibronectin fibers, whereas RFSAFY bound to strained fibers. The probes never attached to the same fiber, which confirmed their ability to selectively discriminate regions within a fibronectin fiber network.
"This study strongly suggests that fibronectin fibers under strain display markedly different biochemical signatures that can be used for the molecular-level detection of fibronectin fiber strain," explained Barker. "The data also show the potential for living tissue to be interrogated for mechano-chemical alterations that lead to physiological and pathological progression."
In the future, the researchers hope to use these fibronectin strain-sensitive probes to target therapeutics to fibronectin fibers based on their mechanical signature.

















