Cause of rare growth disease discovered
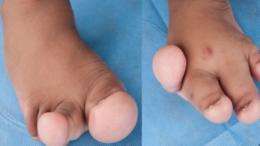
A rare genetic disease which causes some parts of the body to grow excessively has been linked to a cancer-associated mutation that drives cell growth, potentially paving the way for new treatments. The research findings were published in Nature Genetics.
An international collaboration among the University of Cambridge, the Sanger Institute, and the Babraham Institute in the UK and the National Institute for Health in the US has discovered that unrestrained and sometimes massive fatty ‘overgrowth’ affecting only some body regions is caused by mutations in the phosphatidylinositol-3-kinase (PI3K)/AKT signalling pathway (which is critical for cellular growth and metabolism).
The types of mutations which cause these overgrowths typically arise during embryonic development. Unlike conditions caused by genetic mutations which are transmitted from parents (in which every cell in the body is affected), in this condition only the ‘offspring’ of the cell where the mutation occurs carry the change. This accounts for why only some parts of the body are affected.
The scientists from the University of Cambridge and the Sanger Institute, who were funded mainly by the Wellcome Trust, first studied a patient who had severe overgrowth of her legs but a normal upper body. By conducting a genetic analysis of cells sampled from the affected and unaffected areas, they were able to identify a critical mutation which caused the overgrowth in her legs. Scientists at the Babraham Institute then made use of their unique lipidomics mass spectrometry capability to demonstrate abnormal activation of the signalling pathway in cells from the leg but not the arm. These studies were extended in collaboration with scientists at the US National Institute for Health in Maryland, leading to discovery of nine other patients with similar mutations, confirming the results and suggesting that activation of the PI3K pathway may be a common cause of this form of fatty overgrowth.
Dr Robert Semple, from the University of Cambridge Metabolic Research Laboratories at the Institute of Metabolic Science, said: “The mutations we have found are well known as ‘cancer mutations’. However, in cancers they are found with numerous other genetic changes, while here they are apparently in isolation.”
“Studying our patients will thus give new information about the specific role played by these mutations in cancer. Moreover, the major effort to make drugs targeting these mutations for cancer therapy should benefit people with this rare problem, and when drugs safe enough for long-term use are developed they may offer for the first time a targeted, and effective, non-surgical treatment for the excessive growth. We are testing currently available drugs in cells at present.”
Dr Inȇs Barroso, from the Wellcome Trust Sanger Institute, said “We wanted to understand the biology behind this rare and debilitating disorder and thought exome sequencing – sequencing the region of the genome where genetic material is translated into proteins – would be a powerful approach to facilitate the discovery of the underlying mutation. Using DNA sequencing we found that a mutation associated with cancer in the gene PIK3CA was found only in the affected cells of patients”.
Professor Michael Wakelam from the Babraham Institute said “This study exemplifies the importance of collaboration between basic and clinical scientists in translating methodologies and concepts from the research lab to bring about greater understanding and potentially treatment of human disease.”