Some stem cells can trigger tumors
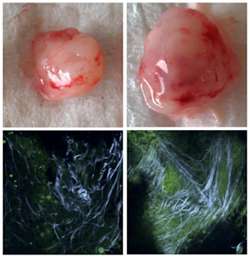
(Medical Xpress) -- Stem cells often used in reconstructive surgery following mastectomies and other cancer-removal treatments may pose a danger: Cornell biomedical scientists have discovered that these cells, in contact with even trace amounts of cancer cells, can create a microenvironment suitable for more tumors to grow.
"It is necessary for us not only to think about what happens with these cells in an otherwise healthy patient, but also, what the fate of stem cells may be in a patient who is prone to disease," said Claudia Fischbach-Teschl, assistant professor of biomedical engineering, who led the research published in Proceedings of the National Academy of Sciences, June 4.
The cells the researchers studied are derived from fat and are called adipose-derived stem cells. They are useful for tissue engineering and reconstructive surgery because they are good at taking over healthy tissue function and recruiting new blood vessels to promote healing.
But they might be too good -- that is, the Cornell researchers observed that the presence of cancer cell media -- the soluble material that contains chemicals secreted by tumor cells -- prevents the stem cells from turning into fat cells as would be desired. Instead, they triggered the cells to secrete chemicals known as "factors" that promote blood vessel growth, or angiogenesis, and to develop into myofibroblasts, which are cells known to play a role in tumor development.
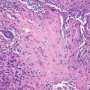
These alterations led to a stiffening of the extracellular matrix that surrounds the cells -- the stiffening is a characteristic feature of breast cancer (which is why tumors can be palpated). Myofibroblasts make the surrounding tissue more rigid, and this stiffness triggers more changes in the stem cell behavior that lead to even more tumor-promoting characteristics -- a positive feedback loop.
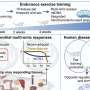
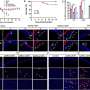
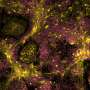
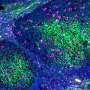
The researchers observed these changes in in vitro experiments using stem cells and breast cancer cell lines that varied in aggression. First they collected soluble media from tumor cells and observed how the stem cells changed in response. They found that TGF-beta and interleukin-8 are specific tumor-secreted factors that contribute to the stem cells' eventual change in phenotype to myofibroblasts. They confirmed their results with in vivo experiments by injecting stem cells and tumor cells into the mammary glands of mice.
The experimental results are also supported by the fact that obese women are more likely to develop breast cancer. The presence of more adipose tissue means larger numbers of adipose stem cells, and one could hypothesize that the larger stem cell pool could promote tumor-progression processes, Fischbach-Teschl said.
The paper, whose first authors are graduate students Emily Chandler and Bo Ri Seo, resulted from a multidisciplinary collaboration that included researchers from the College of Engineering, College of Veterinary Medicine and College of Arts and Sciences.
The research was supported by the National Institutes of Health, Cornell Center on the Microenvironment and Metastasis and the National Science Foundation.