Breakthrough treatment reduces post-surgical scarring for glaucoma patients
(Medical Xpress) -- Scientists at the Singapore Eye Research Institute (SERI) and Nanyang Technological University (NTU) have developed an innovative way to combat post-surgical scarring for glaucoma patients.
A clinical trial has shown that the use of a new drug delivery method has resulted in 40 per cent fewer injections needed by glaucoma patients to prevent scarring after surgery. This also means fewer hospital visits for these patients in future.
Glaucoma, a disease characterised by a build-up of pressure in the eye, is a major cause of blindness worldwide. It affects about 3 per cent of the population in Singapore and an estimated 30 per cent of sufferers require surgery to adequately control the disease. However, success rates for glaucoma surgery in Asian patients are considerably lower than those reported in Caucasian patients because Asians have a higher risk of scarring after such surgery. Up to one out of three operated patients requires a minor surgical procedure in the first six months in order to maintain the ideal low post-operative eye pressure.
“The post-operative scarring response is the major obstacle for successful glaucoma surgery. We’ve seen in our clinics that Asian patients scar earlier and more aggressively than their Caucasian counterparts, and a significant number require at least one post-operative intervention to treat this scarring response,” said Associate Professor Tina Wong, Senior Consultant with SNEC’s Glaucoma Service, and Head of the Ocular Therapeutics and Drug Delivery Research Group at SERI. She is also the senior author of this study.
The breakthrough treatment method is made possible by Professor Subbu Venkatraman, Acting Chair of NTU’s School of Materials Science and Engineering, who invented a way to make the drug, which prevents post-surgical scarring, last longer at the site of the injection. This considerably increases the interval before the drug has to be administered again.
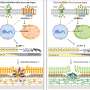
Using a gel know as hyaluronic acid, Professor Venkatraman discovered a way to contain the drug, 5-Fluorouracil (5-FU) inside the gel. “Leveraging NTU’s expertise in controlled-release technology, we have found a way to deliver the drug 5-Fluorouracil gradually into the patient. This allows the drug to be time-released over several days, compared to the current effect of the drug which remains at the injected site for only a few hours. The benefit for patients who have undergone glaucoma surgery is clear – fewer injections of the drug are needed. This results in less post-surgical scarring and fewer visits to the hospital.”
The aim of glaucoma surgery is to create a new pathway for the aqueous fluid to flow out, thereby lowering eye pressure. During glaucoma surgery, a flap is created using the patient’s own tissue to regulate the outflow of aqueous fluid. A small blister, known as a conjunctival bleb, marks the new surgically created filtration site.
The flap that allows fluid to flow out can be blocked if scar tissue forms, preventing the fluid from draining effectively and causing the eye pressure to rise again. This is a particularly common problem in Asian patients. To restore outflow through the surgical flap, the obstructing scar tissue needs to be removed by injecting 5-FU which prevents further scarring. This procedure is called bleb needling.*
The clinical trial involved 49 patients, who were randomised to receive an injection of either the current 5-FU solution or the new combined formulation following bleb needling. All subjects were followed up for three months. The trial was conducted at the Singapore National Eye Centre.
The team found that the subjects who were randomised to receive the new treatment had an improved post-operative outcome. “With this novel treatment, we observed a dramatically lower rate for repeat needling, with only 12 per cent requiring further intervention, whereas 50 per cent of subjects receiving the standard 5-FU solution treatment required further needlings,” said Dr Arun Kumar Narayanaswamy, Senior Clinical Research Fellow, SERI, and first author of this study.
“In addition, because these patients require fewer interventions, their risk of ocular infection and side effects are significantly reduced,” he added.
Assoc Prof Wong, also an adjunct professor at NTU’s School of Materials Science and Engineering, and Professor Subbu Venkatraman together with his team of scientists, are improving the new treatment method further using nano-encapsulation. The team aim to achieve a precise release of the correct amount of drug at a steady daily dose over a course of several weeks instead of just a few days as shown by the study.
“Because the acute and most active stage of wound healing occurs in the first twelve weeks after surgery, we ideally need a sustained time release of the anti-scarring drug that can be administered as a single injection and provide the right amount of drug to continually suppress the scarring response for that crucial time frame. That way, we won’t have to keep injecting patients with top-ups, often on a fortnightly or even weekly basis which is not only inconvenient for the patient but greatly increases the risk of complications with each additional injection,” said Assoc Prof Wong.
In the future, this novel treatment could also be applied at the time of the glaucoma surgery to further improve surgical outcomes, as well as reduce the possible need for or frequency of bleb needling interventions after surgery.
The study, published in the journal Ophthalmology early this year, has been shortlisted for a Best Clinical Research Oral Presentation at the second SingHealth Duke-NUS Scientific Congress, which will take place on 3 and 4 August at Raffles City Convention Centre.
This research was supported in part by an Individual Research Grant awarded to Assoc Prof Wong by the Singapore Ministry of Health’s National Medical Research Council.