Study: Gene-gene interactions important to trait variance
(Medical Xpress)—Gaining more insight into predicting how genes affect physical or behavioral traits by charting the genotype-phenotype map holds promise to speed discoveries in personalized medicine. But figuring out exactly how genes interact has left parts of the map invisible.
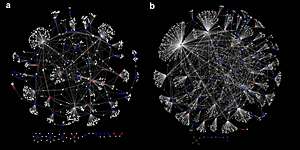
In a paper published in Proceedings of the National Academy of Sciences, North Carolina State University genetics researchers reveal some of the hidden portions of the map, showing that complicated networks of gene-gene interactions in fruit flies greatly influence the variance in quantitative traits, or characteristics that are influenced by multiple genes – like sensitivity to alcohol or aggression.
The effects of these gene-gene interactions, known as epistatic effects, are difficult to gauge in human populations because some variations are unknown, says Dr. Trudy Mackay, William Neal Reynolds and Distinguished University Professor of Genetics at NC State and the paper's corresponding author.
"One of the ways we model the genotype-phenotype map in humans – one variant at a time – may be wrong," Mackay says. "Over the past four or five years, thousands of genomewide studies have focused on how variants affect traits, but the effects shown have been really small.
"It's like the parable of the elephant and the blind men, in which one blind man feels the elephant's trunk and believes he's touching a snake, while another man touches the elephant's knee and believes he's touching a tree. Using just one interaction doesn't help the men identify the elephant."
Enter Mackay and her fruit flies – nearly 200 lines of genetically identical flies that are also a sample of genetic variations for different traits, as well as thousands of flies that were crossbred to shuffle up the genes.
In three studied traits, the PNAS paper compared identical flies with crossbred "mutt" flies and, surprisingly, showed no matched variations in chemical building blocks known as single-nucleotide polymorphisms.
However, the paper showed that the epistatic networks could be used to predict variation in the three studied traits.
Mackay says the knowledge gained in this paper could be used to look for epistatic interactions in human genome association studies.
"Understanding how genetic variation in an individual gives rise to susceptibility to complex diseases like diabetes or schizophrenia – or even to traits like height – is one of the open questions in quantitative genetics today, " Mackay says. "Knowing a person's particular variants could motivate therapeutic treatments."
The study was funded by the National Institutes of Health and the National Human Genome Research Institute. Colleagues from the Baylor College of Medicine collaborated on the research.
More information: Epistasis Dominates the Genetic Architecture of Drosophila Quantitative Traits, Trudy F.C. Mackay, et al. Published online the week of Sept. 3, 2012 in Proceedings of the National Academy of Sciences
Abstract
Epistasis – non-linear genetic interactions between polymorphic loci – is the genetic basis of canalization and speciation, and epistatic interactions can be used to infer genetic networks affecting quantitative traits. However, the role epistasis plays in the genetic architecture of quantitative traits is controversial. Here, we compared the genetic architecture of three Drosophila life history traits in the sequenced inbred lines of the Drosophila melanogaster Genetic Reference Panel (DGRP) and in a large outbred, advanced intercross population derived from 40 DGRP lines ("Flyland"). We assessed allele frequency changes between pools of individuals at the extremes of the distribution for each trait in the Flyland population by deep DNA sequencing. The genetic architecture of all traits was highly polygenic in both analyses. Surprisingly, none of the single nucleotide polymorphisms (SNPs) associated with the traits in Flyland replicated in the DGRP, and vice versa. However, the majority of these SNPs participated in at least one epistatic interaction in the DGRP. Despite apparent additive effects at largely distinct loci in the two populations, the epistatic interactions perturbed common, biologically plausible, and highly connected genetic networks. Our analysis underscores the importance of epistasis as a principal factor that determines variation for quantitative traits and provides a means to uncover novel genetic networks affecting these traits. Knowledge of epistatic networks will contribute to our understanding of the genetic basis of evolutionarily and clinically important traits and enhance predictive ability at an individualized level in medicine and agriculture.
















