Study uncovers molecular role of gene linked to blood vessel formation
University of North Carolina researchers have discovered that disrupting a gene that acts as a regulatory switch to turn on other genes can keep blood vessels from forming and developing properly.
Further study of this gene – a "transcription factor" called CASZ1 – may uncover a regulatory network that influences the development of cardiovascular disease. A number of other studies have already shown a genetic link between mutations in CASZ1 and hypertension.
The UNC research, which was carried out in a frog model as well as human cells, will be published April 29, 2013, in the journal Developmental Cell.
"There has been a lot of interest in studying the vasculature because of its role in a wide range of disease states, as well as human development. But there are very few transcription factors that are known to affect the vasculature. To find a new one is quite unique, and then to be able to link it up to a known network of vascular development is surprising and encouraging," said senior study author Frank Conlon, PhD, an associate professor of genetics in the UNC School of Medicine.
During vascular development, specialized cells coalesce into three-dimensional "cords" that then hollow out to provide a path for transporting blood throughout the body. This process involves the complex coordination of molecular entities like growth factors and signaling molecules, defects that have been associated with human illnesses such as cancer, stroke, and atherosclerosis.
Conlon has long been interested in understanding how these various molecular players come together in the cardiovascular system. In 2008, his laboratory showed that a gene called CASZ1 is involved in the development of heart muscle. In this study, he and his colleagues decided to look for its role in the development of blood vessels.
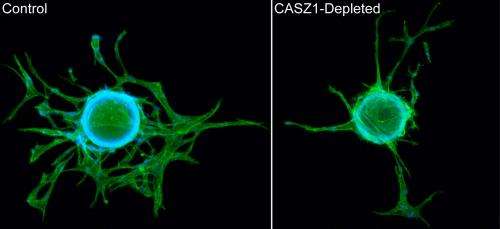
Marta S. Charpentier and Kathleen S. Christine, lead authors of the study and graduate students in Conlon's laboratory, removed CASZ1 from frog embryos and looked to see how its absence affected the development of the vasculature. Without CASZ1, the frogs failed to form branched and functional blood vessels. When they removed the CASZ1 gene from cultured human cells, Charpentier and Christine saw similar defects: the cells did not sprout or branch correctly due to their inability to maintain proper adhesions with the surrounding extracellular matrix.
"If you take out CASZ1, these cultured human cells try to migrate by sending out these filopodia or little feet, but what happens is it is like someone nails down the back end of those growing vessels. They try to move and keep getting thinner and thinner, and like an elastic band it gets to be too much and just snaps back. It appears to cause an adhesion defect that makes the cells too sticky to form normal vessels," said Conlon.
CASZ1 is a transcription factor, a master switch that controls when and where other genes are expressed. Therefore, Charpentier and Christine did a series of experiments to explore CASZ1's influence on a known vascular network, involving other genes called Egfl7 and RhoA. When Charpentier and Christine added the Egfl7 gene to her CASZ1-depleted cells, the defect in blood vessel formation went away, suggesting that the two genes are connected. They then showed that CASZ1 directly acts on the Egfl7 gene, and that this activity in turn activates the RhoA gene, which is known to be required for cellular behaviors associated with adhesion and migration.
Transcription factors themselves are so essential that they are generally considered to be "undruggable," but the researchers say that further studies into how specific transcription factors work and the targets they control could eventually lead to new drug candidates.
"Egfl7 is a therapeutic target of interest, because companies such as Genentech are already working on it for cancer therapy," said Charpentier. "Figuring out how it is regulated is important not just for understanding the biology of it, but also for discovering targets that could trigger the development of innovative therapeutic strategies for cardiovascular disease."