November 6, 2013 report
An artificial blood substitute from Transylvania
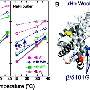
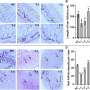
(Medical Xpress)—Researchers in Cluj-Napoca, Romania, have recently made some significant advances in developing artificial blood substitutes. Their formulation is based not on synthetic hemoglobins, but rather on hemerythrin protein extracted from marine worms. Led by Professor Radu Silanghi-Dumitrescu, from Babes-Bolyai University, the team has been testing their blood substitute in both mice and in cultured cells. Their initial results suggest that many of the adverse effects normally associated with either perfluorocarbon (PFC) or hemoglobin-based oxygen carrier (HBOC) substitutes can be eliminated, or at least minimized by using hemerythrin.
Human blood only has a shelf life of a few weeks. It also needs to be matched to the recipient's blood type, and while the risks of disease transmission can be minimized by testing, dangers still present if the donor has been recently infected. These facts of life have lead to huge efforts to build a better blood substitute, but so far results have not been encouraging.
What makes hemoglobin such a success, is a phenomenon known as cooperative binding. In a nutshell, as more oxygen binds to hemoglobin in the lungs, it becomes even easier for additional oxygen to bind. Similar affects also seem to occur in the opposite direction, for the unloading of oxygen, in tissues. Most hemerythrins are only about one-quarter as efficient as hemogobin, mainly because cooperative binding is not as common with them.
The problem with HBOCs is that one way or another, the hemoglobin they contain ends up escaping and causing serious damage to organs like the kidneys. HBOCs are usually only around 0.1 um in diameter, compared to 7um for red blood cells (RBCs). While their small size allows them to penetrate and oxygenate the nooks and crannies of the body much better than RBCs, that same feature also leads to undesireable extravasation into tissue. The typical stop gap is to try to make the hemoglobin in HBOCs sturdier by cross-linking, polymerizing, or otherwise conjugating them to various polymers.
Researchers have found when these modified hemoglobins do get into tissues, they bind nitric oxide, which appears to have the result that the patient's blood pressure rises precipitously. HBOCs also cause cramping and abdominal pains which are likely the result of oxidative/nitrosative free-radical generation. The new tipple from Transylvania, which instead uses the hemerythrin, has much lower free-radical stress related reactivity than hemoglobin. The researchers looked at these effects both in human leukocytes and human umbilical cells. When compared with standard glutaraldehyde-polymerized bovine hemoglobin, their new substitute resulted in much healthier cultures.
The goal is not to develop a permanent replacement solution, but rather something that could be used to bridge a critical situation for the few hours or days it takes for the body's natural mercenaries to take over. Hemerythrin itself, despite its name, is not actually a heme molecule in the strict sense. It is virtually colorless when deoxygenated, but when bound with oxygen it turns a violet-pink. It is interesting to speculate if these new blood substitutes, or something similar to them, might one day afford new opportunity to image live brains in formerly unimagined detail.
Aside from scattering of light by lipids, heme in blood is also a major problem for optical imaging of the brain. Temporary use of blood substitutes may eventually permit new techniques similar to the Clarity methods to be used in live brains. Hemerythrin has many other unique properties which still need to be explored. Among its positive features, for example, are a much reduced affinity for carbon monoxide. While there will no doubt be many obstacles yet to be overcome before something like this could be put into widespread use, the results to date are very encouraging.
More information: www.ias.ac.in/jbiosci/jun2011/215.pdf
© 2013 Medical Xpress