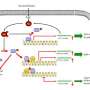
Two potential therapies for preventing myofibroblast differentiation
Multiple types of cells are needed for patients to recover from surgery or to restore life to tissues that have lost circulation. University of Georgia researchers have identified how the cells involved in the final stage of repair, myofibroblasts, are switched on. This information can lead to drug development that could speed recovery.
Injuries heal when inflammatory cells remove dead tissues, and new blood vessels form to supply nutrients for new tissue to grow. Another necessary step in the healing process is the addition of fibroblasts, cells that build the structural framework to connect tissues. If this final stage of repair, called the fibroblast-mediated repair, becomes uncontrolled it can lead to tissue fibrosis.
"Fibrotic scars formed in the brain after a stroke and myocardial infarction in the heart are major roadblocks for recovery," said Somanath Shenoy, associate professor with the UGA College of Pharmacy.
Other examples of fibrosis include alcohol- and hepatitis-induced liver fibrosis, stiffening of blood vessels during pulmonary arterial hypertension and a dangerous clinical condition called idiopathic pulmonary fibrosis, which has no approved treatment except for a lung transplant.
Shenoy and his colleagues at the UGA Center for Pharmacy and Experimental Therapeutics led a recent study looking into pathological significance of Akt activity in the generation of myofibroblasts, which is an underlying cause of fibrotic diseases. Akt is a specific protein kinase responsible for sugar metabolism, cell migration and transcription.
"In our study, we focused on the molecular mechanisms regulating the 'switch' from physiological fibroblasts to pathological myofibroblasts, and we identified a new signaling pathway in fibroblasts that regulates this pathologic switch, which is an Akt kinase-mediated pathway involving transcription factors Serum response factor and myocardin," Shenoy said. "We used biochemical, genetic, transgenic mouse and a translational approach to test our hypothesis and characterize the role of Akt-SRF-myocardin signaling axis in myofibroblast differentiation."
The results of the study are available at www.jbc.org/content/288/46/33483 .
The study tested two potential therapies for preventing myofibroblast differentiation: an Akt inhibitor and an SRF inhibitor.
Previous work by Shenoy has shown transient activation of the Akt pathway by fibroblast growth factor is necessary for tissue recovery following a wound and ischemia tissue injury. The current study identified for the first time that sustained and uncontrolled Akt activation is integral for myofibroblast differentiation and tissue fibrosis.
The study also tested three compounds-triciribine (Akt inhibitor), rapamycin (mTOR inhibitor) and CCG1423 (SRF inhibitor)-for their therapeutic benefits in preventing myofibroblast differentiation. The medications were tested for their ability to reverse the switch from pathological myofibroblasts to more normal fibroblasts.
"The inhibition of Akt by triciribine will blunt the transforming growth factor beta-induced myofibroblast differentiation," said Maha Abdalla, lead author of the paper and doctoral candidate in Shenoy's laboratory. "The effect of hypoxia on myofibroblast formation and pulmonary artery remodeling was blunted in mice lacking the Akt gene."
"Altogether, we identified the novel mechanisms regulating tissue fibrosis and demonstrated the potential therapeutic benefits of triciribine (an Akt inhibitor) for fibrotic disorders such as hepatic and idiopathic pulmonary fibrosis," Abdalla said.
The study was funded by UGA College of Pharmacy dean's special endowment funds, American Heart Association grant #0830326N and National Institutes of Health grant #R01HL103952.
The research team is currently investigating the therapeutic potential of Akt inhibitors for the treatment of pulmonary hypertension using a mouse model.