January 14, 2014 report
Researchers find coevolution of Heliobacter pylori strains has affected gastric cancer risk
(Medical Xpress)—Gastric adenocarcinoma, a form of stomach cancer, is the second most common cause of death from cancer, accounting for 10 percent of all cancer deaths. The bacterium Helicobacter pylori, found in the gastric mucosa of more than half the people in the world, causes gastric adenocarcinoma. However, less than one percent of those who harbor the bacterium develop this cancer. To understand why the risk is greater in some than in others, Barbara Schneider of Vanderbilt University Medical Center in Nashville and her colleagues studied two geographically close populations with a similar prevalence of H. pylori but very different rates of gastric adenocarcinoma. They found that when H. pylori strains have had a long time to coevolve along with their human hosts, there is a reduced risk of gastric adenocarcinoma. The research appears in the Proceedings of the National Academy of Sciences.
Schneider and her team knew that in Central and South America, people who live on the coast are more likely to develop gastric cancer and precancerous gastric stomach lesions than those who live in the mountains. They wanted to see if genetic differences between coastal and mountain people and between the H. pylori they carried accounted for the difference in risk.
The researchers studied residents of two towns in Colombia: Tuquerres in the mountains and Tumaco on the coast. Although the two towns are only 200 kilometers apart, and both have about a 90 percent rate of H. pylori infection, the risk of gastric adenocarcinoma is 25 times as high among people in Tuquerres as it is among people in Tumaco.
When the team examined the subjects' DNA and their gastric mucosa, they found the Tuquerres population to be mostly of Amerindian ancestry. However, the people of Tuquerres were more likely to harbor strains of H. pylori from Europe than from East Asia, where Amerindians originated. Schneider's team think Spanish conquistadores who arrived in the Americas in the 15th century brought these European strains with them. On the other hand, the people of Tumaco, mostly descendants of African slaves, were predominantly of African ancestry. Their gut mucosa mostly contained African H. pylori strains.
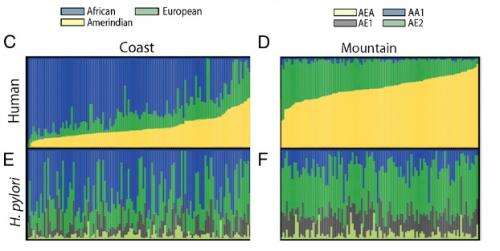
Schneider's team think the African strains found in the Tumaco population coevolved with their human hosts, gradually becoming less virulent. Over time, pathogens tend to become less dangerous to their hosts. The European strains, introduced to the Tuquerres population only recently, have had much less time to coevolve. Therefore, they are much more dangerous to the people of Tuquerres who carry them.
More information: Human and Helicobacter pylori coevolution shapes the risk of gastric disease, PNAS, www.pnas.org/content/early/2014/01/08/1318093111
Abstract
Helicobacter pylori is the principal cause of gastric cancer, the second leading cause of cancer mortality worldwide. However, H. pylori prevalence generally does not predict cancer incidence. To determine whether coevolution between host and pathogen influences disease risk, we examined the association between the severity of gastric lesions and patterns of genomic variation in matched human and H. pylori samples. Patients were recruited from two geographically distinct Colombian populations with significantly different incidences of gastric cancer, but virtually identical prevalence of H. pylori infection. All H. pylori isolates contained the genetic signatures of multiple ancestries, with an ancestral African cluster predominating in a low-risk, coastal population and a European cluster in a high-risk, mountain population. The human ancestry of the biopsied individuals also varied with geography, with mostly African ancestry in the coastal region (58%), and mostly Amerindian ancestry in the mountain region (67%). The interaction between the host and pathogen ancestries completely accounted for the difference in the severity of gastric lesions in the two regions of Colombia. In particular, African H. pylori ancestry was relatively benign in humans of African ancestry but was deleterious in individuals with substantial Amerindian ancestry. Thus, coevolution likely modulated disease risk, and the disruption of coevolved human and H. pylori genomes can explain the high incidence of gastric disease in the mountain population.
© 2014 Medical Xpress