New simple way to reset cells could be transplant "game changer"
Scientists Wednesday reported a simple way to turn animal cells back to a youthful, neutral state, a feat hailed as a "game-changer" in the quest to grow transplant tissue in the lab.
The research, reported in the journal Nature, could be the third great advance in stem cells—a futuristic field that aims to reverse Alzheimer's, cancer and other crippling or lethal diseases.
The latest breakthrough comes from Japan, as did its predecessor which earned its inventor a Nobel Prize.
The new approach—provided it overcomes safety hurdles—could smash cost and technical barriers in stem-cell research, said independent commentators.
"If it works in man, this could be the game-changer that ultimately makes a wide range of cell therapies available using the patient's own cells as starting material," said Chris
Mason, a professor of regenerative medicine at University College London.
"The age of personalised medicine will have arrived."
Stem cells are primitive cells that, as they grow, differentiate into the various specialised cells that make up the different organs—the brain, the heart, kidney and so on.
The goal is to create stem cells in the lab and nudge them to grow into these differentiated cells, thus replenishing organs damaged by disease or accident.

One of the obstacles, though, is ensuring that these transplanted cells are not attacked as alien by the body's immune system.
To achieve that, the stem cells would have to carry the patient's own genetic code, to identify them as friendly.
In 1998 came the first gain: the use of cloning technology—pioneered with Dolly the sheep—to harvest stem cells from early-stage embryos grown from the donor's own DNA.
Hugely versatile, these "pluripotent" stem cells are controversial as the method entails destroying the embryo, something opposed by religious conservatives and others.
In 2006, a team led by Shinya Yamanaka of Kyoto University, who was a co-recipient of the 2012 Nobel Prize for Medicine, created so-called induced pluripotent stem cells (iPS).

With this, the team took mature cells and coded them with four genes, "rewinding" the cells' genetic programmes to return them to a juvenile state.
The technique had to overcome an early hurdle of causing tumours in cells and still faces problems with efficiency—less than one percent of adult cells typically are reprogrammed successfully.
The latest breakthrough, pioneered by Haruko Obokata at the RIKEN Center for Developmental Biology in Kobe, takes an entirely different and surprisingly low-tech approach.
'Hallmarks of pluripotency'
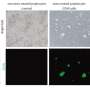
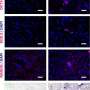
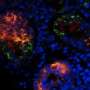
White blood cells in newborn mice were returned to a versatile state by incubating them in a solution with high acidity for 25 minutes, followed by a five minute spin in a centrifuge and a seven-day spell of immersion in a growth culture.
Called stimulus-triggered acquisition of pluripotency (STAP) cells, the innovation breaks new ground.
Until now, only plant cells, and not mammal cells, have been found to reprogramme back to a youthful state through simple environmental factors.
"These STAP cells show all the hallmarks of pluripotency," Obokata said Tuesday in an Internet briefing with journalists.
STAP cells appear to have a limited ability to self-renew, and it has yet to be seen if they can be acquired from humans.
Only much later may trials in humans follow.
It took 13 years before the first trials with embryonic stem cells, and six years before the first iPS trials.
Dusko Ilic, a stem-cell scientist at Kings College London, said the approach "is indeed revolutionary."
But he cautioned: "It does not bring stem cell-based therapy closer."
"We will need to use the same precautions for the cells generated in this way as for the cells isolated from embryos or reprogrammed with a standard method," he said in comments reported by London's Science Media Centre.
More information: phys.org/news/2014-01-embryoni … em-cells-embryo.html
© 2014 AFP