May 1, 2014 report
Fast contractions and depolarizations in mitochondria revealed with multiparametric imaging
(Medical Xpress)—When something bad happens to otherwise healthy neurons it's easy to blame the usual suspects—the mitochondria. In some cases the nucleus might be the one at fault, as in a de novo mutation in a critical gene or in some other runaway error process in the instruction pipeline. Othertimes there could be leakage into the brain of toxins, bacteria, or even overzealous patriot cells of the host. But by and large, it's the mitochondria who bear responsibility for nearly everything the brain does and so it is they who must accept it when it fails. To better understand how these organelles function, researchers have turned to special imaging methods that let them observe multiple aspects of their behavior all at once.
In one of the most revealing studies of its kind to date, researchers in Germany were able to observe the tiny contractions that mitochondria undergo during their complex shifts through different redox states and levels of depolarization. Publishing in a recent issue of Nature Medicine they relate these effects to pH and calcium concentration in the both the mitochondria and surrounding axon, and also to the larger spiking activity of the neuron.
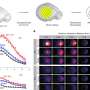
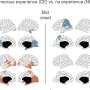
Led by Martin Kerschensteiner and Thomas Misgeld, the group was able to bring together several new intracellular imaging agents, and imaging instruments, under the same experimental setup. In particular, they transgenically expressed a unique biosensor for glutathione redox potential in neuronal mitochondria. This sensor goes by the rather convoluted name of glutaredoxin-1 (Grx1)–reduction-oxidation sensitive GFP2 (roGFP2). They were able to obtain both high temporal resolution, and subcellular spatial resolution, of the redox state of this biosensor using excitation-ratiometric confocal and two-photon imaging. The calcium, pH, and membrane potential changes were also simultaneously observed in both in normal mice, and in ASL or spinal cord injured mice.
Researchers have been eager to look beyond single-variable relationships and instead focus on the structure-function relationships that emerge under collective observation. In other words, not just changes in intensity, but changes in configuration. Here researchers could track not only shape changes in mitochondria themsleves, but also the movements, and therefore locations of these organelles. The researchers didn't find, at least under physiologic conditions, any major links of glutathione redox potential to organelle location or movement. However, individually, they found spontaneous contractions in the mitochondria. These reversible contractions were amplified when neurons were stimulated, and also when they were stressed either acutely or chronically.
Under multiparametric imaging the researchers were able to piece together a more general biochemical scenario that relates the contractions to respiratory chain–dependent depolarization of the membrane potential. Energetic uncoupling, as well as alkalinization of the matrix compartment of the mitochondria then follow these events. (In biochem-speak, uncoupling generally refers the normal proton gradient in mitochondria that either collapses or is no longer linked to ATP synthesis.) It was also found that when axons were stressed, particularly under full axotomy, these mitochondrial changes were preceded by a rise in axonal calcium. This increase propagated down the axon at twice the speed of the associated redox change and ultimately induced a sharp increase in mitochondrial calcium.
The frequency of mitochondrial contractions, at least those large enough to be observed, was only around 0.6 per hour. One wouldn't say that contractions are ongoing in the sense that the action potentials that neurons fire are ongoing. But with something on the order of 10,000 mitochondria per cell it would seem that it might always be "contraction time" somewhere in the cell. Assuming that is, that the mitochondria are independent from each other, and from any synchronizing influence of the neuron itself. If there is some synchronizing influence that is observed though, certaining the spike of a neuron would be a good candidate for it.
Other researchers are also beginning to recognize the importance of these issues for the cell. In a recent related study done at Georgia Regents University, researchers looked for any signs of simultaneous behavior in the filamentous mitochondria of hepatocytes. They were able to show that transient contractions in mitochondria induce membrane depolarizations through a mechanism involving an inner membrane dynamin protein known as OPA1. Using a potentiometric probe known as TMRE, they found that flickering in the membrane potential occurs in a compartmentalized fashion, but that not all mitochondria in cell act together.
For now, it is difficult to match these slower contractions, calcium sparks, and redox potentials with faster events, like spikes in the neuron. As we mentioned before, perhaps the most useful genetically-encoded sensors will be those that can indicate rapid, but small temperature changes. Temperature, which can even be measured through the course of a neuron's spike, could emerge as the one variable by which many others are compared.
More information: Multiparametric optical analysis of mitochondrial redox signals during neuronal physiology and pathology in vivo, Nature Medicine (2014) DOI: 10.1038/nm.3520
Abstract
Mitochondrial redox signals have a central role in neuronal physiology and disease. Here we describe a new optical approach to measure fast redox signals with single-organelle resolution in living mice that express genetically encoded redox biosensors in their neuronal mitochondria. Moreover, we demonstrate how parallel measurements with several biosensors can integrate these redox signals into a comprehensive characterization of mitochondrial function. This approach revealed that axonal mitochondria undergo spontaneous 'contractions' that are accompanied by reversible redox changes. These contractions are amplified by neuronal activity and acute or chronic neuronal insults. Multiparametric imaging reveals that contractions constitute respiratory chain–dependent episodes of depolarization coinciding with matrix alkalinization, followed by uncoupling. In contrast, permanent mitochondrial damage after spinal cord injury depends on calcium influx and mitochondrial permeability transition. Thus, our approach allows us to identify heterogeneity among physiological and pathological redox signals, correlate such signals to functional and structural organelle dynamics and dissect the underlying mechanisms.
© 2014 Medical Xpress