Researchers use world's smallest scaffolding to build new livers
A UK company is using technology originally designed for use in Space to assist in a ground-breaking project that aims to improve the treatment of liver-associated diseases. Ultimately it could develop a new generation of liver replacements, using fibres a hundred times thinner than a human hair.
Waiting for a replacement organ that never comes is the heart breaking truth for 14% of patients requiring a liver transplant. The demand for replacement livers has always far outweighed supply, with over 10,000 people waiting for a liver transplant in the EU alone. Many patients die, or become too sick to undergo surgery during the wait.
Now, the Electrospinning Company (TECL), a spin out from the Science and Technology Facilities Council (STFC), is taking part in Re-Liver, a ground-breaking EU funded project in regenerative medicine, to improve the treatment of liver-associated diseases and, ultimately, develop a new generation of liver replacements.
With industrial and academic partners in Germany and Italy, alongside the University of Manchester, the project is developing artificially grown, mini liver organs, known as organoids. These organoids consist of cells that provide liver cell functions, which could be used to treat liver-associated diseases such as haemophilia.
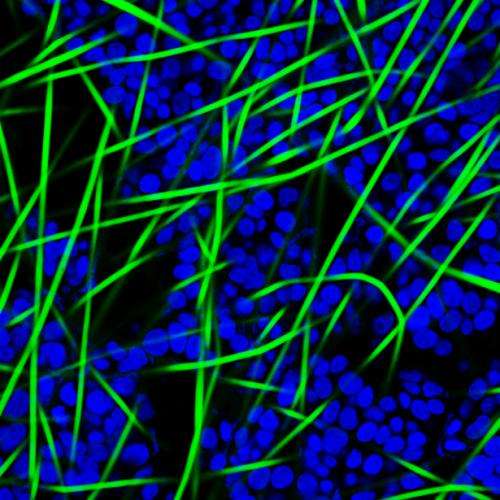
For its part in the project, TECL is combining space technology developed at STFC with a process known as electrospinning, in which an electrical charge is used to produce fibres that are a hundred times thinner than a human hair. These fibres are electrospun into microscopic 3D scaffolds, much smaller than the eye can see. Composed from synthetic, medical grade polymers which mimic the cellular behaviour of real human tissues, these scaffolds have been approved by the US Food and Drug Administration (FDA).

Dr Kate Ronayne, Head of Innovation at STFC, said: "TECL's microscopic scaffolds provide the ideal environment to support the growth of functioning 3D liver cells. Its role in the project could have a huge impact on the future of regenerative medicine. This is a fantastic example of how world class science, teamed with STFC's business management and innovation support, can help small companies, with brilliant ideas, to address the really significant challenges facing our society today."
This three year project will confirm proof of principle for this technology, and investigate all safety aspects, to allow clinical trials to begin. The long-term goal is the development of artificial organoids that could be transplanted into the body to supplement, or even eventually replace, lost liver activity.
Ann Kramer, CEO of TECL, said: "We are thrilled to be part of this project, which has the real potential to impact both regenerative medicine and liver toxicity screening in the near future. Our participation has been helped greatly by the innovation management and support of STFC."