August 7, 2014 report
The fastest neurons in the brain
(Medical Xpress)—If you ask a child which is the world's fastest animal, they may tell you anything from cheetah, falcon or swift, to sailfish. If you ask a neuroscientist what is the fastest spiking neuron, you probably won't even get an answer right away. A few might offer that the neurons that form the auditory nerve can track a sound waveform with spikes at up to 1000hz—for a little while at least. Others might note that cells in several brainstem complexes can blast away at over half that speed for much longer periods of time when stabilizing the eyeballs. But if you continue to press them, they might respond with the familiar manta: "fast-spiking parvalbumin-positive interneurons"
If parvalbumin (a calcium-binding protein found in muscle and synapses) and fast-spiking interneurons sound like strange bedfellows to you, you are not alone. So what then exactly is this protein speed pill, and is its strange association with measures of global performance in cells a mere quirk of the path of biology through history, or an inevitable neurotruth bound to have been discovered sooner or later? A recent review, published in Science takes a close look at the unique properties of these particular neurons as individuals, and explores their community behavior within the context of the rhythms and oscillations in larger circuits.
The main deal with these particular interneurons is that whether they are the so-called chandelier cells in the cortex, Purkinje cells in the cerebellum, or basket cells in the hippocampus, they all use the transmitter gaba. Neuroscientists gabba lot about gaba. If you browse through the poster sessions at one of their large conferences you can actually hear the background hum of hundreds of gaba murmurings falling in and out of phase with each other. It sounds something like this: gabba, gabba, gabba, gabba, gabba........
The review details the common sense concept that fast neurons need fast parts. Stated another way, a neuron can only be as fast as its slowest link in the signal chain. In the traditional conception, the fastest neuron in the world can only transfer spike-based information as fast and reliably as subsequent vesicle fusions at the synapse can track them. However, if fusion probabilities are only 50 percent at best for spike rates of just a few hz, this idea quickly gets into trouble as spike rates increase. We would expect these fusion probabilities at individual synapses to fall precipitiously for rates a hundred times that. As we know from other studies, at least in vitro, it is not too hard to make neurons run out of transmitter.
Many of the basket-type parvalbumin cells send out short axons which redundantly envelope the cell bodies of other neurons. Conceivably, at high spike rates, these massive terminations could be activated "round robin" so to speak, like the rotor inside a distributor cap which fires each piston in sequence. In this way high spike rates could activate different synapses at different times to deliver a more-or-less reliable signal to a particular neuron. Without the synaptic endurance afforded by the elaborate vesicle ribbons and rapid fire cartridges that certain sensory neurons possess, mere interneurons could make due by just using extra wiring. One thing that the authors note, however, is that the majority of parvalbumin interneurons are not just local basket cells. Many send out axons of significant length that branch diffuse regions contacted hundreds of neurons.
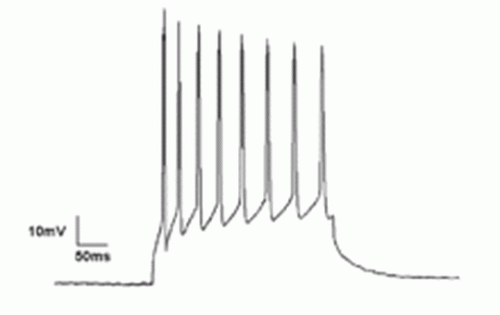
Researchers have found that the presynapses of these neurons use highly optimized isoforms of standard synaptic proteins and channels. In addition to parvalbumin, they employ synaptotagmin II, which happens to have the fastest calcium binding kinetics out of the 15 or so different varieties of this protein. And it's not just the synapses, these neurons are first class all the way. Only the fastest activating and deactivated calcium and potassium channels from the most elite families are tolerated (for the physiologists, this means none of those pokey L or N type channels). Additionally these neurons have among the highest sodium channel densities found for unmyelinated axons. While they may only be able to send spikes at a relatively modest 1.5 m/s, more importantly, they fire the tightest spikes in the brain.
Parvalbumin is more than just calcium kinetics. It is fairly abundant in the tissues where it is found and it happens to be the protein responsible for seafood allergy—and for that matter, frog leg allergy. The parvalbumin (and a few other closely related proteins like troponin) in the fast-contracting muscles of these animals is slightly different from our own. In those afflicted this can provoke a strong IgE antibody reaction. What happens to parvalbumin after its binds calcium is still a bit of a mystery. Similar proteins, like calmodulin for example, undergo conformational changes after calcium binding to expose hydrophobic methyl groups on its methionine residues. These surfaces bind a wide variety of targets, particularly those that contain amphiphilc helices with complimentary hydrophobic domains. In parvalbumin interneuron synapses, it has been found that release only need a few calcium channels to operate. The local positions and conformations of parvalbumin and other associated proteins may be critical here. Rather than some fluid Brownian machine, we might imagine what we have here, can be better likened to a geared and well-oiled mechanism.
Just how fast are these neurons? That depends. In the absence of the famed oscillatory behavior in the hippocampus, these cells only put out spikes at around 6 Hz. During theta rhythm (4-10Hz) they might jump up to 20 Hz, while during gamma rhythm and sharp wave ripple, they fire over 120 Hz. That is certainly below the single millisecond scale bursts of an auditory neuron, but still not too shabby.
Hot rodders know the fastest car is not the one with the biggest engine or most expensive components. It is the one whose parts work best together. As in racing, swapping out stock proteins for racing proteins is just the first step—they must then be tuned together in an evolutionary trial and error. Parvalbumin is likely just one of many components that must work smoothly together for maximal performance.
More information: Fast-spiking, parvalbumin+ GABAergic interneurons: From cellular design to microcircuit function, Science 1 August 2014: Vol. 345 no. 6196. DOI: 10.1126/science.1255263
© 2014 Medical Xpress