New pathway for stalling BRCA tumor growth revealed
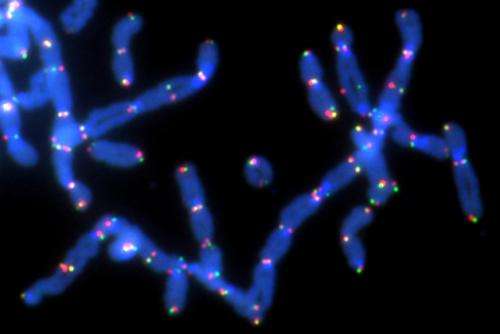
Inhibiting the action of a particular enzyme dramatically slows the growth of tumor cells tied to BRCA1 and BRCA2 genetic mutations which, in turn, are closely tied to breast and ovarian cancers, according to researchers at NYU Langone Medical Center.
Senior investigator and NYU Langone cell biologist Agnel Sfeir, PhD, says that if further experiments prove successful, these findings could lead to a new class of targeted therapies against cancers with BRCA1 and BRCA2 mutations. The researchers' findings in experiments in mice and human cells are described in the journal Nature online Feb. 2.
Dr. Sfeir and her collaborators say their discovery about the enzyme—called polymerase theta, or PolQ—resulted from efforts to answer a fundamental biological question: How do cells prevent the telomere ends of linear chromosomes, which house our genetic material, from sticking together? Cell DNA repair mechanisms can stitch together telomeres broken as part of cell metabolism. But such fusions, the researchers say, compromise normal cell growth and survival.
"In the purest biological sense, our findings show how this particular enzyme, which we know is active in several tumors, promotes unwanted telomere fusions by inserting whole segments of DNA via a disruptive DNA repair pathway termed alt-NHEJ," says Dr. Sfeir, an assistant professor at NYU Langone and its Skirball Institute for Biomolecular Medicine and a member of NYU's Laura and Isaac Perlmutter Cancer Center. "It was quite remarkable to find that by blocking PolQ action, cancer cell growth was cut by more than half."
For the study, Dr. Sfeir and colleagues at the Scripps Research Institute focused their analysis on telomeric DNA. They noted that as the chromosome ends were being joined, whole sections of new genetic material were being inserted into the telomeric DNA—suggesting that any of a dozen or more DNA-synthesizing polymerase enzymes were at work. Researchers then focused on PolQ, in part because it is known to be active in several tumors, including breast and ovarian, but also in liver and colon cancers. Breast and ovarian cancers are among the leading causes of cancer death among women in the United States.
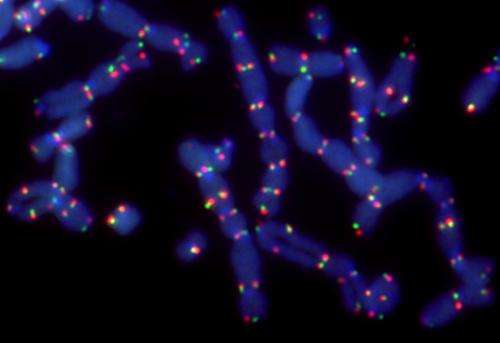
Additional experiments confirmed that PolQ is needed to activate the alt-NHEJ pathway of DNA repair. Unlike the main, error-free pathway—or HDR pathway—the alt-NHEJ pathway does not use a related chromosome's genetic material as a template to meticulously correct any damaged genetic material. As such, alt-NHEJ is highly likely to leave coding mistakes.
"Our studies will continue to look at how the alt-NHEJ pathway operates," Dr. Sfeir adds, "and what biological factors cells use in addition to PolQ to choose between the error-prone or error-free DNA repair pathways."
More information: Nature, DOI: 10.1038/nature14157" target="_blank">nature.com/articles/DOI: 10.1038/nature14157