February 5, 2016 report
Researchers making progress on understanding why people lose their hair as they age

(Medical Xpress)—Two groups of researchers working on two different projects have made inroads into understanding the cause of alopecia—the gradual loss of hair during aging. One team, with members from Japan, the U.S. and the Netherlands, found that accumulated DNA damage was at least partly to blame—the other team, working at the University of Colorado, discovered a certain protein that appears to be responsible for causing follicle stem cells to enter their dormant cycle. Both teams have published papers in the journal Science, describing their studies and results. Mingxing Lei and Cheng-Ming Chuong with the University of Southern California offer a Perspectives piece on the work done by the two teams in the same journal issue, and more fully explain the on/off growing cycle of scalp hair in mammals.
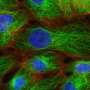
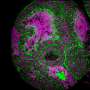
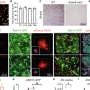
Understanding why people lose their hair as they age might benefit people looking to avoid baldness, but it might also offer a whole new understanding of the way that stem cells work. In one of the two studies, the researchers sought to find the answer to a question that plagues dermatologists and other researchers—is alopecia caused by environmental factors, or is it because of some internal mechanism? The team was not able to prove definitively that it is likely more due to the latter, but found strong evidence that suggests the root cause is accumulated damage to DNA over the course of many cell mutations. To come to that conclusions they took skin samples from aged mice and found DNA damage had led to the breakdown of a protein called Collagen 17A1 and that led to follicle stem cells being transformed into a type of skin cell, which eventually got sloughed off. To find out if the same applied to humans, the researchers studied hair follicles taken from a large group of women between the ages 22 and 70. In addition to lower levels of Collagen 17A1, they also found that follicles were smaller in the older women.
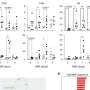
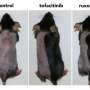
To better understand the hair cycle, the second team of researchers focused on follicle stem cells during both phases of hair growth—the growing (anagen) and resting (telogen) phases—and in so doing they found that a protein called Foxc1 appeared to be the trigger for keeping hair growth dormant—when the gene that caused it to be expressed was removed, the follicles stayed in the "on" phase indefinitely. Not a cure for baldness, of course, but certainly an interesting discovery.
More information: 1. H. Matsumura et al. Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis, Science (2016). DOI: 10.1126/science.aad4395
Abstract
Hair thinning and loss are prominent aging phenotypes but have an unknown mechanism. We show that hair follicle stem cell (HFSC) aging causes the stepwise miniaturization of hair follicles and eventual hair loss in wild-type mice and in humans. In vivo fate analysis of HFSCs revealed that the DNA damage response in HFSCs causes proteolysis of type XVII collagen (COL17A1/BP180), a critical molecule for HFSC maintenance, to trigger HFSC aging, characterized by the loss of stemness signatures and by epidermal commitment. Aged HFSCs are cyclically eliminated from the skin through terminal epidermal differentiation, thereby causing hair follicle miniaturization. The aging process can be recapitulated by Col17a1 deficiency and prevented by the forced maintenance of COL17A1 in HFSCs, demonstrating that COL17A1 in HFSCs orchestrates the stem cell–centric aging program of the epithelial mini-organ.
2. L. Wang et al. Foxc1 reinforces quiescence in self-renewing hair follicle stem cells, Science (2016). DOI: 10.1126/science.aad5440
Abstract
Stem cell quiescence preserves the cell reservoir by minimizing cell division over extended periods of time. Self-renewal of quiescent stem cells (SCs) requires the reentry into the cell cycle. In this study, we show that murine hair follicle SCs induce the Foxc1 transcription factor when activated. Deleting Foxc1 in activated, but not quiescent, SCs causes failure of the cells to reestablish quiescence and allows premature activation. Deleting Foxc1 in the SC niche of gene-targeted mice leads to loss of the old hair without impairing quiescence. In self-renewing SCs, Foxc1 activates Nfatc1 and bone morphogenetic protein (BMP) signaling, two key mechanisms that govern quiescence. These findings reveal a dynamic, cell-intrinsic mechanism used by hair follicle SCs to reinforce quiescence upon self-renewal and suggest a unique ability of SCs to maintain cell identity.
© 2016 Medical Xpress