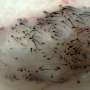
MicroRNAs contribute to hair loss and follicle regression
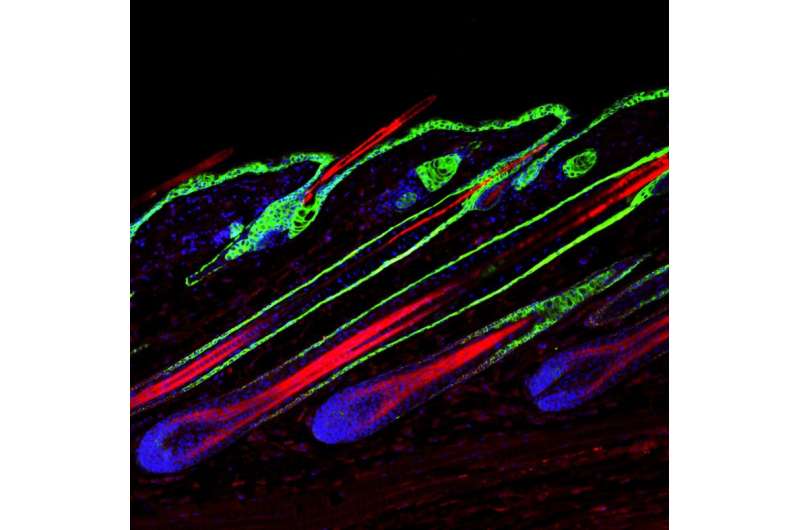
Up to 60% of men experience some degree of hair loss in their lifetime. However, despite its prevalence, efficient treatment for hair loss is lacking. One of the key distinguishing features of hair follicles in baldness-affected areas is premature regression. This leads to shorter hairs and excessive hair fallout. This month's cover image features actively growing hair follicles with prominent layers of the outer root sheath surrounding the hair shaft cortex. In this issue of PLOS Genetics, we describe an essential role for a highly conserved microRNA, miR-22, in regulating the regression of mouse hair follicles. New insights into the mechanism of premature hair growth regression in mice enrich our understanding of the pathogenesis of hair loss.
The basics of hair loss
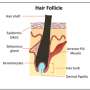
Because hair loss results from the premature termination of the follicle's growth phase, it is essential to understand in more detail the mechanism underlying normal hair regeneration. During the active phase of the hair growth cycle, stem cell activity sustains an actively dividing population of epithelial cells at the base of the follicle called matrix cells. As progeny of the matrix cells move upward from the follicle base (or bulb), they differentiate into a hardened hair shaft, which emerges above the skin surface. Fully differentiated hair shafts consist of dead, but mechanically sound and highly cross-linked, keratin-filled cells. After a period of active hair shaft production, follicles activate an involution program, during which a large portion of epithelial cells die, and the remaining stem cells are reduced to a tight cluster underneath the skin surface. These follicles then remain dormant for some time; however, they can undergo activation and restart active hair shaft production.
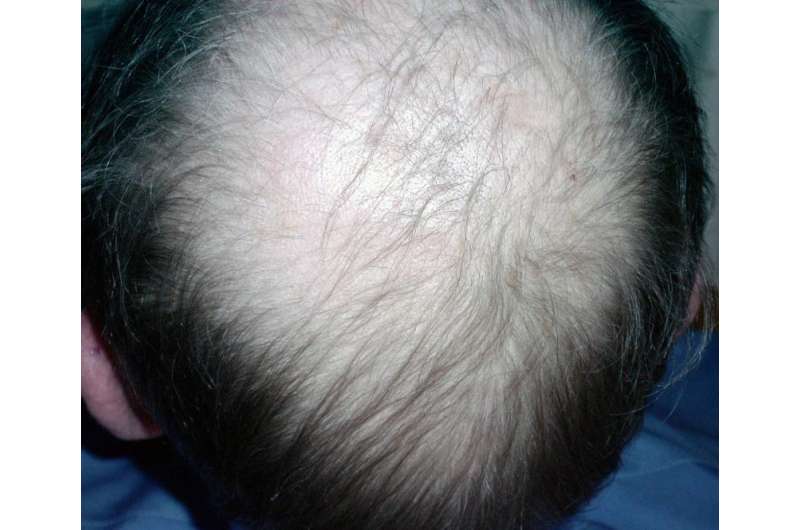
The growth, regression, and resting phases together constitute the hair growth cycle, and this cycling can be influenced by a variety of local and systemic signaling factors. Consequently defects in hair cycling can arise from changes in the normal signaling milieu due to disease, aging, or injury. Commonly, in humans, scalp hair follicles enter resting phase prematurely, and hairs shafts become shorter and fall out, resulting in visible baldness. Therefore, identifying new signaling regulators of hair follicle regression will provide a better understanding of the hair loss pathogenesis mechanism and will likely identify novel therapeutic targets.
miR-22 induction causes premature hair loss by promoting follicle involution
To test the function of miR-22, we generated a genetic tool to induce miR-22 overexpression in mouse hair follicles, and interestingly, found that increasing miR-22 results in hair loss in mice due to the premature regression of actively growing follicles. Surprisingly, our data reveal that the expression of over 50 distinct keratin genes are markedly reduced by miR-22 and that silencing of keratin-mediated hair shaft assembly by miR-22 is a prerequisite for follicle regression. At the molecular level, we found that miR-22 directly represses multiple transcription factors, including Dlx3 and Foxn1, which positively regulate the expression of keratin genes.
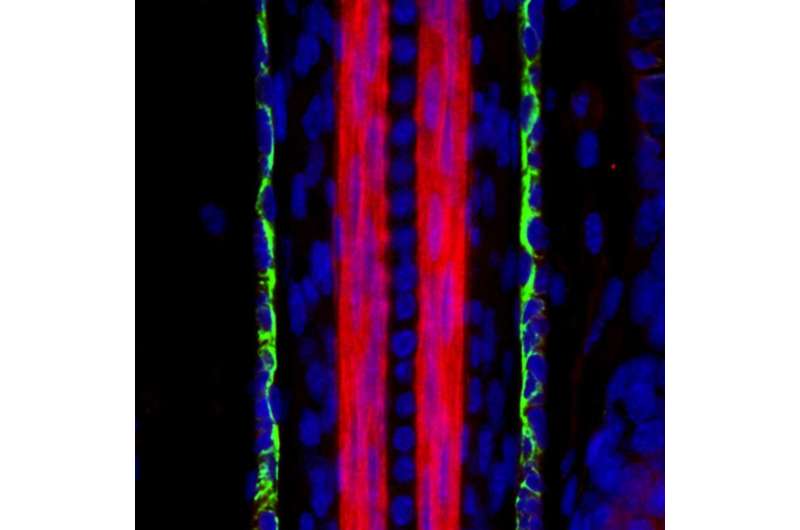
Indeed, deletion of Dlx3 or Foxn1 closely resembles the hair loss phenotype caused by miR-22 induction. Thus, by suppressing Dlx3- and Foxn1-dependent keratin expression, miR-22 is sufficient to terminate hair differentiation. In addition, miR-22 contributes to follicle regression by repressing proliferation of hair stem cells and promoting their death. Collectively, miR-22 emerges as a key regulator of follicle transition from the growth to regression phase. There are hundreds of microRNAs expressed in a hair follicle [2], but most of them are not well studied. Our findings of the essential role of miR-22 highlight the importance of determining the combinatorial effects of the microRNA regulatory network in hair cycling.
Implications of findings
In the future, our findings are likely to benefit human hair loss research efforts. Androgenic alopecia, where premature regression of scalp hair follicles is induced by increasing androgen levels, is the most common hair loss disorder in humans. Interestingly, it has been reported that miR-22 is strongly induced in the liver in response to testosterone treatment [3,4]. Our unpublished data show that two binding sites for an androgen receptor are located in the promoter of both human and mouse miR-22. These findings support the hypothesis that miR-22 functions in the pathogenesis of Androgenic Alopecia, warranting future studies of miR-22 inhibitors as potential anti-hair loss drugs.

More information: 1. Lee J, Tumbar T (2012) Hairy tale of signaling in hair follicle development and cycling. Semin Cell Dev Biol 23: 906-916.
2. Mardaryev AN, Ahmed MI, Vlahov NV, Fessing MY, Gill JH, et al. (2010) Micro-RNA-31 controls hair cycle-associated changes in gene expression programs of the skin and hair follicle. FASEB J 24: 3869-3881.
3. Delic D, Grosser C, Dkhil M, Al-Quraishy S, Wunderlich F (2010) Testosterone-induced upregulation of miRNAs in the female mouse liver. Steroids 75: 998-1004.
4. Wang WL, Chatterjee N, Chittur SV, Welsh J, Tenniswood MP (2011) Effects of 1alpha,25 dihydroxyvitamin D3 and testosterone on miRNA and mRNA expression in LNCaP cells. Mol Cancer 10: 58.