UCSF study will test new vaginal microbicide for herpes and HIV
A team of researchers at UCSF is seeking young women to participate in the first US study of the safety of a new a vaginal gel designed to prevent herpes and HIV infection. If it is effective and approved by the U.S. Food and Drug Administration, the gel would be an important weapon in the fight against HIV because it would allow women to protect themselves from infection rather than relying on their partners to use condoms.
"The significance of the gel is that it potentially gives the power back to the woman to protect herself against infection," said Anna-Barbara Moscicki, MD, professor of pediatrics at UCSF and lead investigator for the study.
The gel is not a contraceptive, but a microbicide. Microbicidal gels or creams are inserted into the vagina solely to prevent the spread of HIV and other sexually transmitted diseases. There are currently no such products on the market. Women who have herpes are at increased risk of contracting HIV, so diminishing the risk of getting herpes also diminishes the risk of HIV infection.
The generic name of the gel is "3 % w/w SPL7013" (brand name VivaGel). Following this safety trial, other trials will be conducted to determine its effectiveness at fighting off herpes and HIV infection. But Moscicki said that trials in animals have found the gel to be nearly 100 percent effective and have few side effects.
"There has been an important move supported by the National Institutes of Health and World Health Organization to support the development of vaginal microbicides," said Moscicki, who also is the director of Teen Clinics in the Division of Adolescent Medicine at UCSF Children’s Hospital. "Herpes infection is the number one attributable cause of HIV infection in the United States and worldwide."
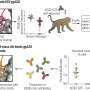
The gel is designed to prevent herpes and HIV infection through the use of a molecule called a dendrimer. Dendrimers have molecular structures that resemble the branches of a tree. The herpes and HIV molecules are "caught" in the dendrimer branches and prevented from entering and infecting human cells, Moscicki said.
To participate in the study, women must be between 18 and 24 years old, sexually active, healthy and free of any sexually transmitted disease. They cannot be pregnant or breast feeding. They must either have regular menstrual periods or not have them at all.
Moscicki said that the safety trial is important because earlier spermicidal microbicides like nonoxynol 9, which was an ingredient in a variety of contraceptive products from condoms to contraceptive creams and gels, was eventually found to increase rather than decrease HIV infection rates.
"Nonoxynol 9 was used commonly and was thought to be an extremely safe type of anti-microbial spermicide. But researchers found that it actually increased the rate of HIV infection in women. So now we realize that we must approach new microbicides a little more carefully," Moscicki said.
According to the U.S. Centers for Disease Control and Prevention, women account for more than one quarter of all new HIV/AIDS diagnoses. Women of color are especially affected by HIV infection and AIDS. In 2002, the most recent year for which data are available, HIV infection was the leading cause of death for African American women aged 25 to 34 years. It was the third leading cause of death for African American women aged 35 to 44 years and the fourth leading cause of death for African American women aged 45–54 years and for Hispanic women aged 35 to 44.
In the same year, HIV infection was the fifth leading cause of death among all women aged 35 to 44 and the sixth leading cause of death among all women aged 25 to 34. The only diseases causing more deaths of women were a variety of different types of cancer and heart disease.
Moscicki said that if the gel is approved by the FDA, women would use it about an hour before having intercourse. "If a woman has occasional sex it could be several times a month. If she were a sex worker it could be several times a day," she said.
The study is being conducted in collaboration with the gel’s manufacturer, Starpharma Holdings, Ltd., of Melbourne, Australia, and is being underwritten by the Sexually Transmitted Infection Clinical Trials Group, which is funded in part by NIH and CDC.
Development of the gel has been given fast-track status by the FDA. Fast track status is granted when a drug has the potential to treat a serious or life-threatening condition and demonstrates the potential to address unmet medical needs for that condition.
Source: University of California - San Francisco