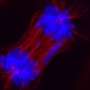
Scientists image a single HIV particle being born
A mapmaker and a mathematician may seem like an unlikely duo, but together they worked out a way to measure longitude – and kept millions of sailors from getting lost at sea. Now, another unlikely duo, a virologist and a biophysicist at Rockefeller University, is making history of their own. By using a specialized microscope that only illuminates the cell’s surface, they have become the first to see, in real time and in plain view, hundreds of thousands of molecules coming together in a living cell to form a single particle of the virus that has, in less than 25 years, claimed more than 25 million lives: HIV.
This work, published in the May 25 advanced online issue of Nature, may not only prove useful in developing treatments for the millions around the globe still living with the lethal virus but the technique created to image its assembly may also change the way scientists think about and approach their own research.
“The use of this technique is almost unlimited,” says Nolwenn Jouvenet, a postdoc who spearheaded this project under the direction of HIV expert Paul Bieniasz and cellular biophysicist Sandy Simon, who has been developing the imaging technique since 1992. “Now that we can actually see a virus being born, it gives us the opportunity to answer previously unanswered questions, not only in virology but in biology in general.”
Unlike a classical microscope, which shines light through a whole cell, the technique called total internal reflection microscopy only illuminates the cell’s surface where HIV assembles. “The result is that you can see, in exquisite detail, only events at the cell surface. You never even illuminate anything inside of the cell so you can focus on what you are interested in seeing the moment it is happening,” says Simon, professor and head of the Laboratory of Cellular Biophysics.
When a beam of light passes through a piece of glass to a cell’s surface, the energy from the light propagates upward, illuminating the entire cell. But when that beam is brought to a steeper angle, the light’s energy reflects off the cell’s surface, illuminating only the events going on at its most outer membrane. By zeroing in at the cell’s surface, the team became the first to document the time it takes for each HIV particle, or virion, to assemble: five to six minutes. “At first, we had no idea whether it would take milliseconds or hours,” says Jouvenet. “We just didn’t know.”
“This is the first time anyone has seen a virus particle being born,” says Bieniasz, who is an associate professor and head of the Laboratory of Retrovirology at Rockefeller and a scientist at the Aaron Diamond AIDS Research Center. “Not just HIV,” he clarifies, “any virus.”
To prove that what they were watching was virus particles assembling at the surface (rather than an already assembled virion coming into their field of view from inside the cell), the group tagged a major viral protein, called the Gag protein, with molecules that fluoresce, but whose color would change as they packed closer together. Although many different components gather to form a single virion, the Gag protein is the only one necessary for assembly. It attaches to the inner face of the cell’s outer membrane and when enough Gag molecules flood an area, they coalesce in a way that spontaneously forms a sphere.
Simon, Bieniasz and Jouvenet found that the Gag molecules are recruited from the inside of the cell and travel to the cell’s surface. When enough Gag molecules get close and start bumping into each other, the cell’s outer membrane starts to bulge outward into a budding virion and then pinches off to form an individual, infectious particle. At this point, the researchers showed that the virion is a lone entity, no longer exchanging resources with the cell. By using tricks from optics and physiology, they were able to watch the steps of viral assembly, budding, and even scission off the cell surface. With such a view they can start to describe the entire lifeline in the birth of the virus.
“I think that you can begin to understand events on a different level if you actually watch them happen instead of inferring that they might occur using other techniques,” says Bieniasz. “This technique and this collaboration made that possible.”
Source: Rockefeller University