MIT zeroes in on Alzheimer's structures
MIT engineers report a new approach to identifying protein structures key to Alzheimer's disease, an important step toward the development of new drugs that could prevent such structures from forming.
In the Aug. 22 issue of PLoS Computational Biology, the researchers describe one such structure uncovered using a new computer-based technique.
Collin M. Stultz, the leader of the work and the W.M. Keck Associate Professor of Biomedical Engineering
in the Department of Electrical Engineering and Computer Science, noted that the same general approach could also be applied to certain proteins associated with cancer.
Alzheimer's disease is the most common form of dementia, affecting some five million Americans, according to the Alzheimer's Association. And due to the growing elderly population, that number "is expected to reach a staggering 13.2 million by 2050," said Stultz, who is also affiliated with the Harvard-MIT Division of Health Sciences and Technology (HST) and MIT's Research Laboratory of Electronics.
Existing therapies, he continued, "do not adequately slow the rate of neurodegeneration in Alzheimer's patients. As such, there is an urgent need to develop new treatments for Alzheimer's dementia."
Stultz's approach to the problem combines his background in engineering and medicine (he holds a PhD and MD, and is a practicing cardiologist with appointments at Brigham and Women's Hospital and the West Roxbury Veterans Administration Hospital). In his work, biochemical experiments inform a novel computer modeling technique aimed at better understanding one type of protein associated with the disease.
Alzheimer's is characterized by two kinds of proteins — amyloid and tau — that aggregate in the brain. Stultz and co-author Austin Huang, an HST graduate student, have focused on determining the structure of tau.
But there's a problem. "Tau is 'natively unfolded,' or floppy, so in solution it moves around a lot and can adopt many different structures," Stultz said, much like the individual strands in a bowl of cooked spaghetti. Contrast that to the vast majority of other proteins, whose individual strands have similar structures, like the individual strands of uncooked spaghetti.
"With a 'normal' protein," Stultz said, "you can measure the lengths of individual molecules and the average will be a pretty good description of any one." Tau molecules, however, "are all over the place — they're so diverse that it's difficult to get one measurement that describes all of the possible structures."
That complicates the hunt for specific tau structures associated with Alzheimer's (not all tau is bad).
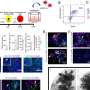
Stultz and Huang approached the problem as engineers, breaking it down into steps. Using a method they developed called Energy-minima Mapping and Weighting (EMW), they asked a computer to come up with all possible structures of tau that are consistent with an average set of experimental data.
"We generated lots and lots of structures for both normal tau and a mutant form" associated with an increased risk for Alzheimer's, Stultz said. By comparing the two sets, the researchers found one structure that was more common in the mutant form — and therefore likely to "play a role in the pathologic process." That structure, in turn, could then become the target for a new drug.
Stultz notes that the current work focused on one tau mutant associated with Alzheimer's, but there are several others. So eventually he hopes to use EMW to create "a list of all types of suspect conformations for known tau mutants. Then, from that list, we can design drugs for each."
Source: Massachusetts Institute of Technology