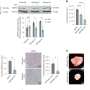
2 drugs are better than 1 at targeting tumors with B-RAF mutations
In a proportion of human solid tumors, in particular melanomas (a form of skin cancer that is often resistant to chemotherapy), inappropriate activation of the MEK/ERK signaling pathway as a result of mutations in the B-RAF gene promotes tumor cell growth and survival. Although MEK inhibitors stop such tumor cells growing, they have a limited ability to kill the tumor cells. Thus, they have had limited success in promoting tumor regression in preclinical and clinical trials.
A team of researchers, at The Walter and Eliza Hall Institute of Medical Research, Australia, has now uncovered the molecular reasons why MEK inhibitors have only a limited ability to kill B-RAF mutant tumor cells and identified another class of drugs that when combined with MEK inhibitors cause tumor regression in mice transplanted with human B-RAF mutant tumor cells.
The team, led by Andreas Strasser and Mark Cragg, found that MEK inhibitors were limited in their ability to kill (by a process known as apoptosis) human B-RAF mutant tumor cells in vitro. The small amount of apoptosis they did induce was mediated via upregulation of the protein Bim. However, if the cells were treated with both a MEK inhibitor and ABT-737 (a drug known as a BH3 mimetic) an extensive amount of apoptosis was observed. Further, the combination also caused tumor regression in mice transplanted with human B-RAF mutant tumor cells; the MEK inhibitor stopped the tumor cells growing and ABT-737 induced the cells to undergo apoptosis. The authors therefore suggest that treating individuals with tumors characterized by B-RAF mutations, especially melanomas, with a MEK inhibitor and a BH3 mimetic might provide a powerful antitumor approach.
Scott Kaufmann and colleagues, at the Mayo Clinic, Rochester, go one step further in their accompanying commentary, asking whether combined MEK inhibitor/BH3 mimetic therapy might be effective for individuals with tumors exhibiting excessive activation of the MEK/ERK signaling pathway in the absence of B-RAF mutations.
Link: www.the-jci.org/article.php?id=35437
Source: Journal of Clinical Investigation