Researchers aim to over-stress already taxed mantle cell lymphoma cells

Cancer cells are already stressed by the fast pace they require to grow and spread and scientists believe a little more stress just may kill them.
"Think about an assembly line in a factory that is working five times faster than normal," said Dr. Kapil Bhalla, director of the Medical College of Georgia Cancer Center. "There is a lot of stress but you need workers to keep going. Some of them fall out, some get bent out of shape."
His research team believes they can disrupt the over-stressed assembly line of mantle cell lymphoma and possibly similar cancers such as pancreatic, liver and breast, by taking away support needed for rapid protein turnover and by clogging up the mechanism for eliminating poorly made ones.
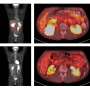
Mantle cell lymphoma, an aggressive cancer of the lymphatic system that mostly occurs in middle age, responds initially to chemotherapy and antibiotics, but often returns, said Dr. Bhalla. Patients have a median survival of three to four years. This cancer affects b lymphocytes, immune cells which make antibodies to fight infection. Ironically, in the process of rearranging genes to make antibodies to a specific invader, mistakes happen, and a would-be protector becomes cancer.
MCG researchers found that to keep their fast pace, these now-malignant cells need increased activity of heat shock protein 90. "Cancer cells require hsp90 for keeping their proteins in active conformation to do their job. That is what cancer is addicted to," said Dr. Bhalla, Cecil F. Whitaker, Jr., M.D./Georgia Research Alliance Eminent Scholar in Cancer and Georgia Cancer Coalition Distinguished Cancer Scholar. Hsp90 is one of the more common molecular chaperones, which help proteins get made, moved, folded and function. Its levels and activity are upregulated in response to stress.
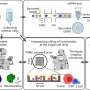
To help push cancer cells over the edge, the researchers are inhibiting hsp90, so the cells lose the molecular chaperone function required to maintain their fast pace. This also puts more stress on the endoplasmic reticulum. Independently hsp90 inhibitors are known to selectively kill cancer cells. But researchers also are clogging up the proteasome, the machinery for chopping up misfolded proteins, recycling some products and eliminating what's left. Much like a sink won't work with a clogged garbage disposal, mantle cell lymphoma cells will start backing up. When a cell detects excessive misfolded proteins, it first has a protective response, but if the problem persists, it commits suicide.
With support from a five-year, $1.5 million grant from the National Cancer Institute, the researchers are using hsp90 and proteasome inhibitors to study protective versus lethal endoplasmic reticulum stress as a way to get rid of mantel cell lymphoma cells. The laboratory studies are being done in human mantle cell lymphoma cells as well as an animal model the researchers developed.
The drugs they are using already are in early clinical trials for a variety of cancers but have not yet been packaged together, Dr. Bhalla said. "We kill cancer cells and a lot of them with this strategy." Still, at least one more inhibitor may get added to the mix. After the rather brutal attack at the cancer's molecular underpinnings, the immune system comes in to essentially mop the floor, but researchers have found cancer cells can still get a pass from an enzyme called IDO. A team of MCG researchers led by Dr. David Munn is exploring IDO's therapeutic potential in cancer. Fetuses use IDO to avoid rejection by the mother's immune system and tumors appear to use it as well. Dr. Bhalla suspects an IDO inhibitor, already under study for lung cancer and other tumors, likely will get a shot at mantle cell lymphoma as well.
Source: Medical College of Georgia